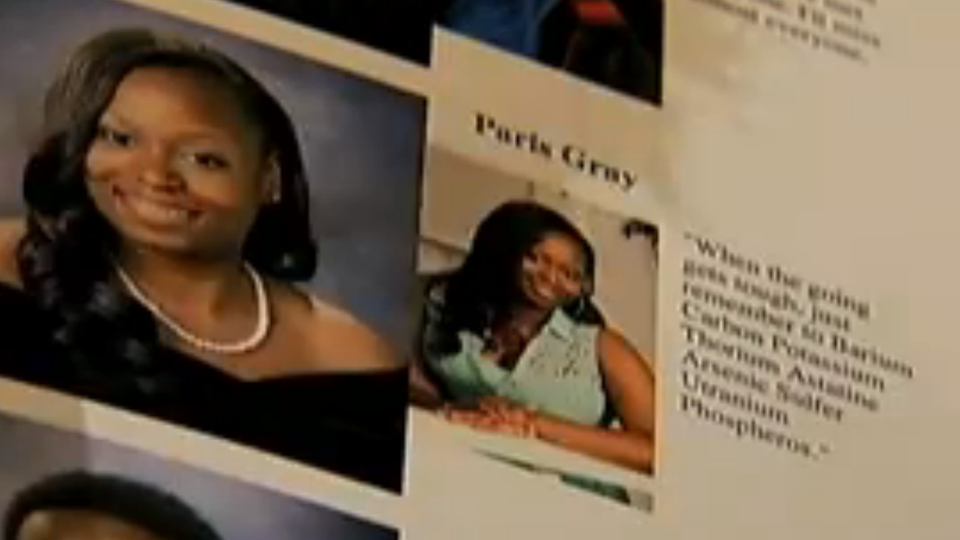Write the balanced chemical equation for the following and identify the type of reaction in each case:(a) Potassium bromide(aq) + Barium iodide(aq) → Potassium iodide(aq) + Barium bromide(s)(b) Zinc carbonate(s) → Zinc

Flourine Uranium Carbon Potassium yttrium oxygen Uranium iodine mytril Adamantium Carbon Astatine - Chemistry Cat - quickmeme

Effect of calcium and barium on potassium-catalyzed gasification of ash-free carbon black - ScienceDirect

Write the balanced chemical equation for the following and identify the type of reaction in each case:(a) Potassium bromide(aq) + Barium iodide(aq) → Potassium iodide(aq) + Barium bromide(s)(b) Zinc carbonate(s) → Zinc
A manual of practical medical electricity : the Röntgen rays and Finsen light . ch it may be united. The following is a list of the commoner elementarysubstances arranged in an electro-chemical

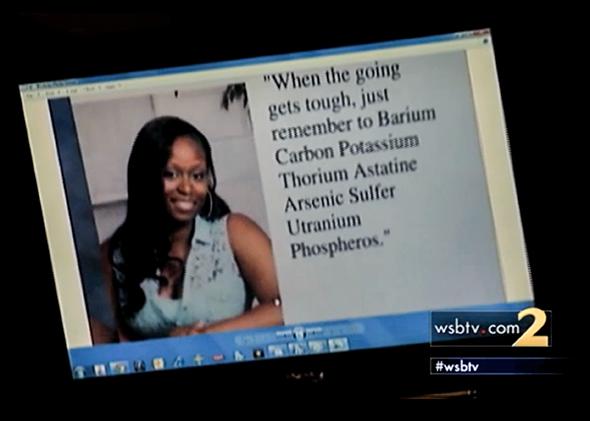
A Periodic Table Yearbook Joke Cost A Student Her Walk At Graduation, Which Doesn't Exactly Make Sense

Question 16: Write balanced chemical equations for reaction.(a) Potassium bromide + barium iodide - YouTube

Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride