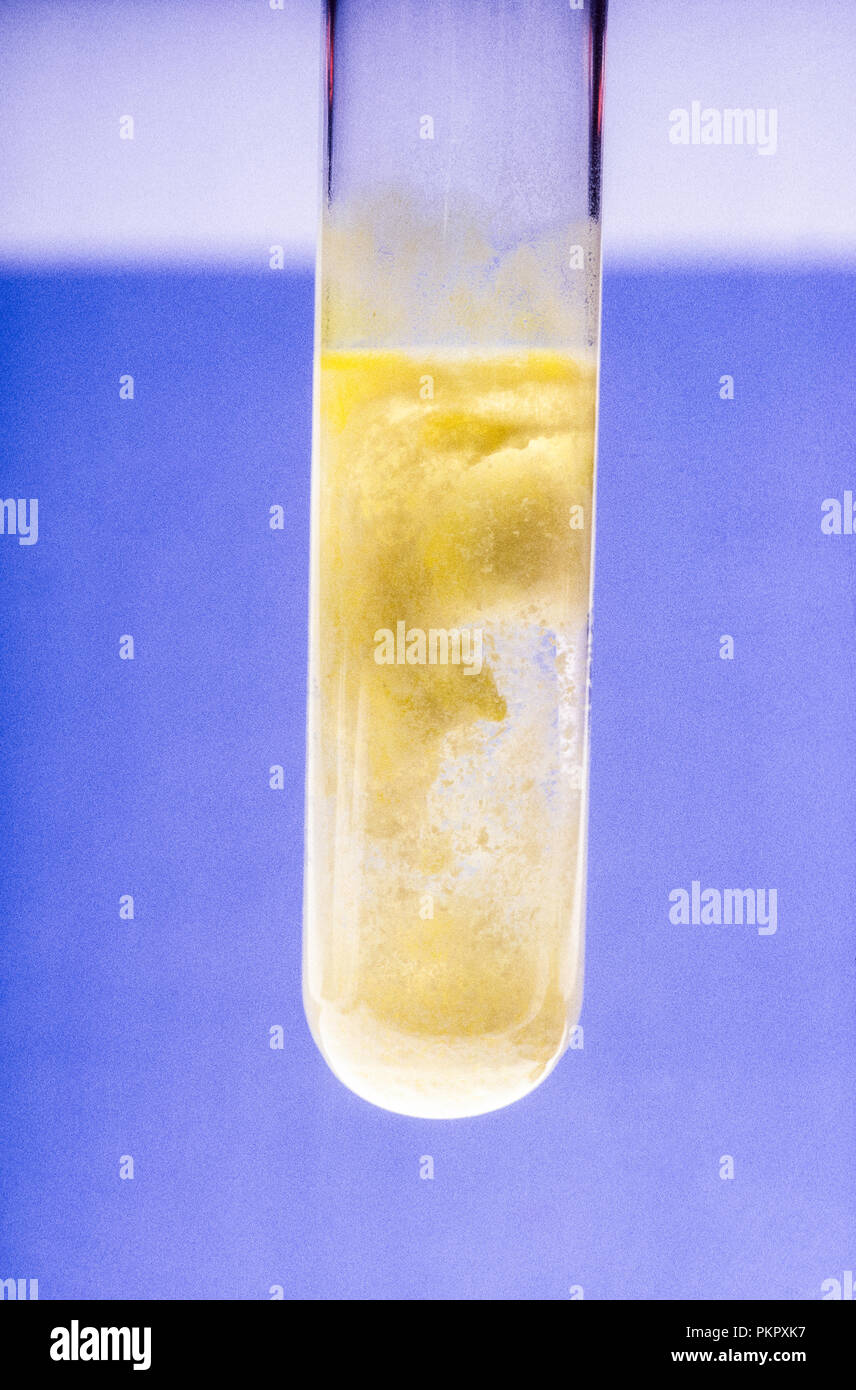
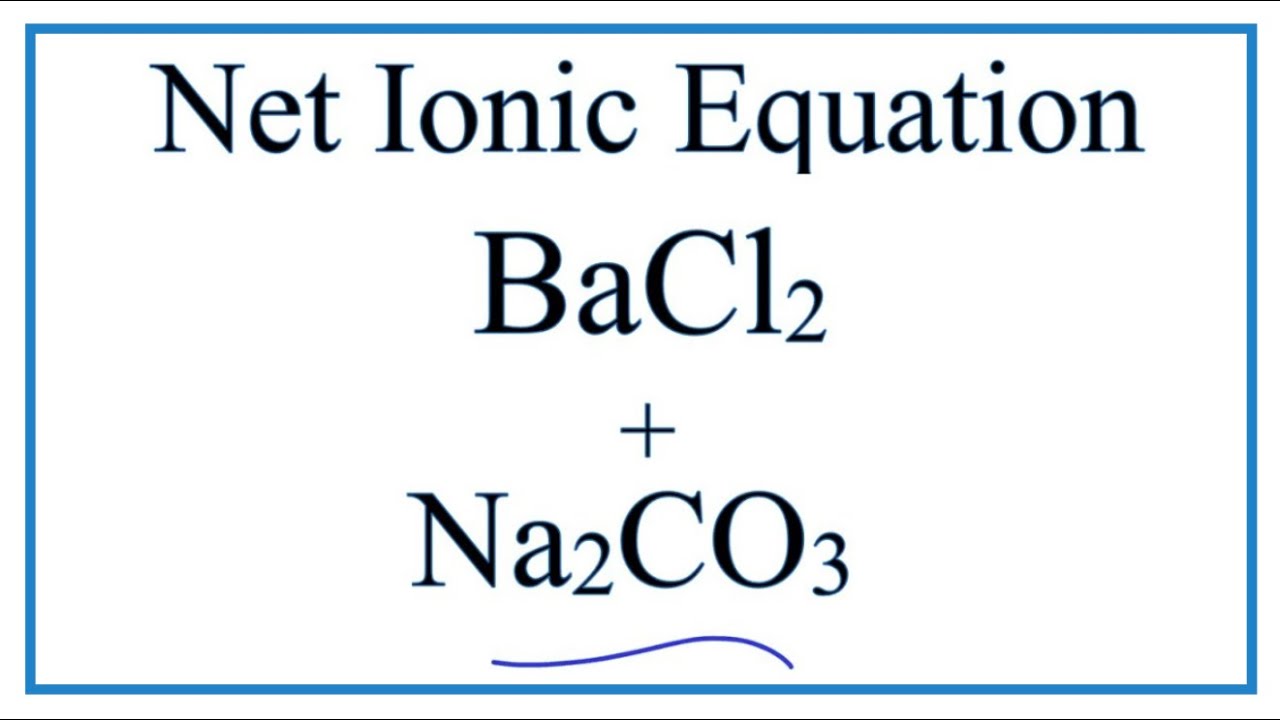SOLVED: I@Lead(II) acetate and sodium carbonate (3Lead(II) acetate and sodium chloride - I4Lead(I) acetate and sodium sulfate SLead(II) acetate and sulfuric acid 'bLead(II) acetate and barium chloride I7Lead(II) acetate and hydrochloric acid
BaCl2 + Na2CO3 - barium chloride and sodium carbonate (double replacement precipitation reaction) - video Dailymotion

Net Ionic Equation for BaCl2 + Na2CO3 = BaCO3 + NaCl | Net Ionic Equation for BaCl2 + Na2CO3 = BaCO3 + NaCl This is an example of a double displacement reaction.

Unit 3 – Chemical Reactions. In many ionic reactions, not all of the chemical species undergo a change. Those that do not change are called spectator. - ppt download

Double displacement of Na2CO3 + BaCl2 | Sodium carbonate + Barium chloride | Precipitation reaction - YouTube

Write the formulae of the salts given below.Potassium sulphate, sodium sulphate, calcium sulphate, magnesium sulphate, copper sulphate, sodium chloride, sodium nitrate, sodium carbonate and ammonium chloride.