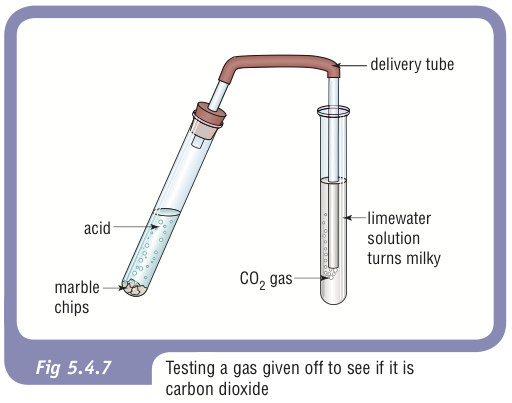
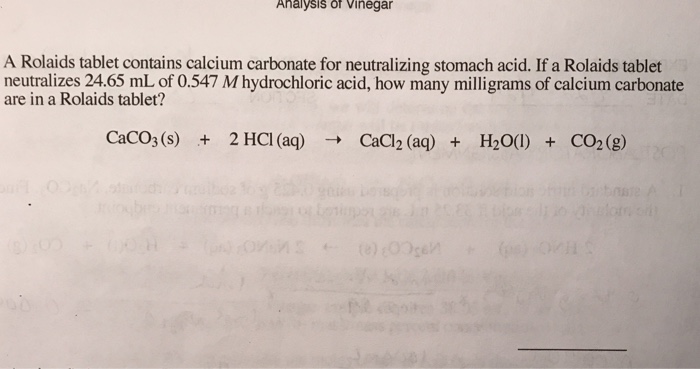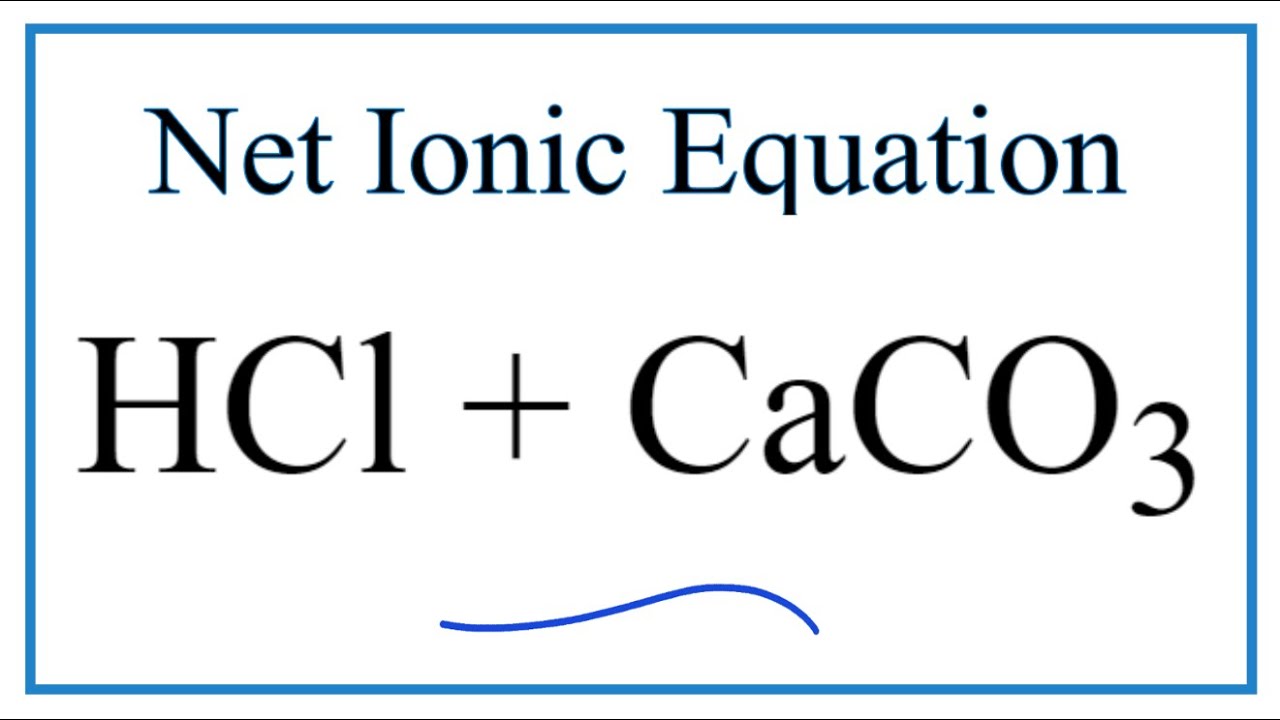calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide Stock Photo - Alamy

SOLVED: Queson 2 Calcium carbonate reacts with hydrochloric acid CaClz(s) according CO2(g) to the following equation: HzOn) CaCO3(s) HClg) Which of the following represents the equilibrium constant expression for the above reaction? [

In an experiment, hydrochloric acid was added to calcium carbonate. These are the equations I came up with. Can somebody check them for me? Additionally, I'm thinking the net ionic equation is

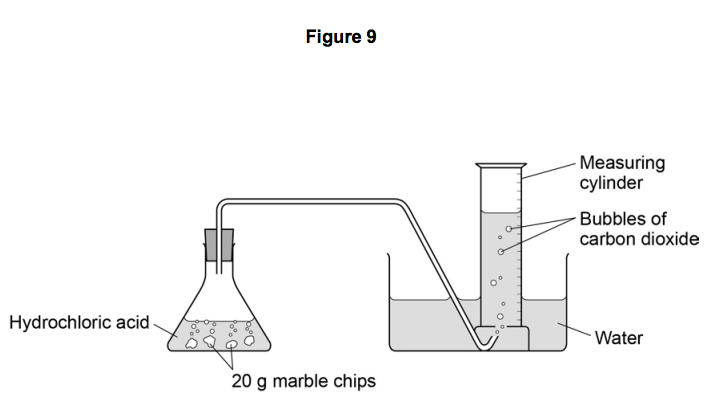
P A student carried out an experiment to investigate the rate of reaction between powdered calcium carbonate and excess dilute hydrochloric acid, using the following apparatus. The student measured the mass of

calcium carbonate reacts with dilute hydrochloric acid to produce carbon dioxide Stock Photo - Alamy

reaction mechanism - Calcium carbonate and hydrochloric acid - data analysis - Chemistry Stack Exchange

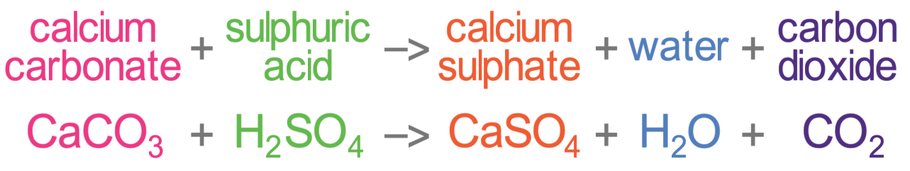
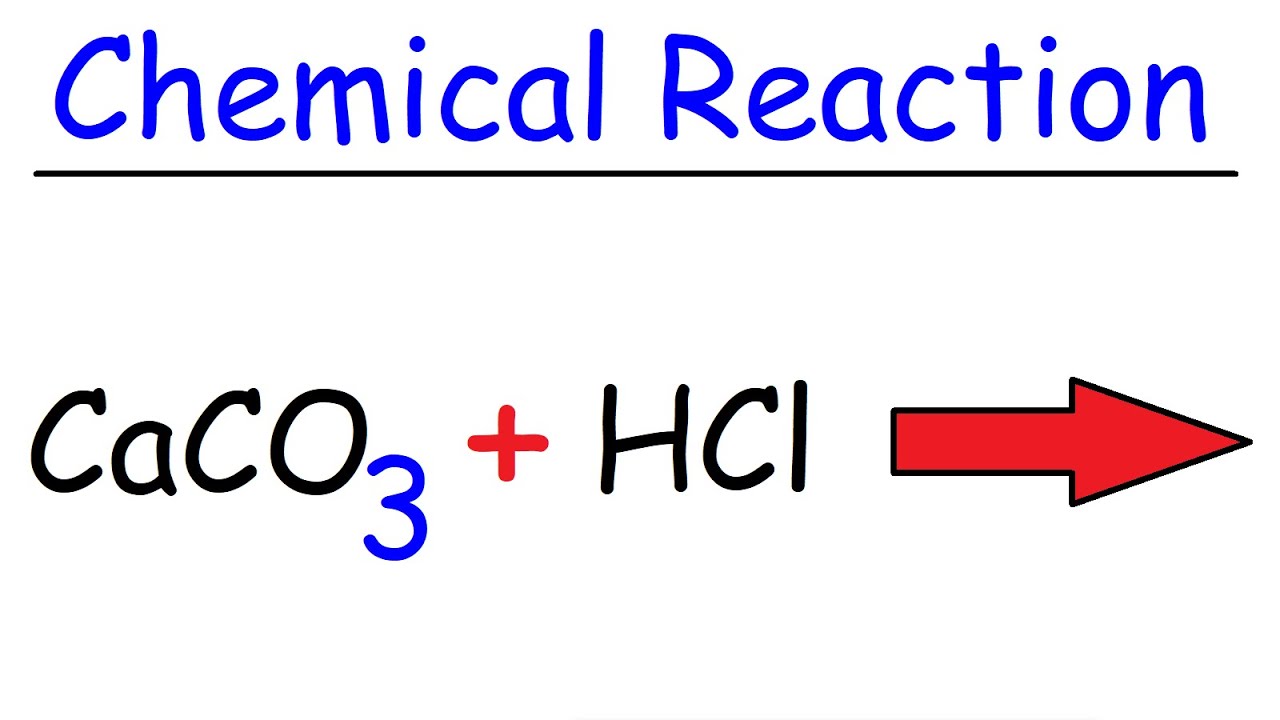
Write a balanced chemical equation for the reaction of calcium carbonate and dil. hydrochloric acid.

![MCQ] - Which correctly represents Parent acid and base of Calcium MCQ] - Which correctly represents Parent acid and base of Calcium](https://d1avenlh0i1xmr.cloudfront.net/0014c0c3-c848-4073-a814-6e71c0e2bf5e/reaction-to-form-calcium-carbonate---teachoo-01.jpg)