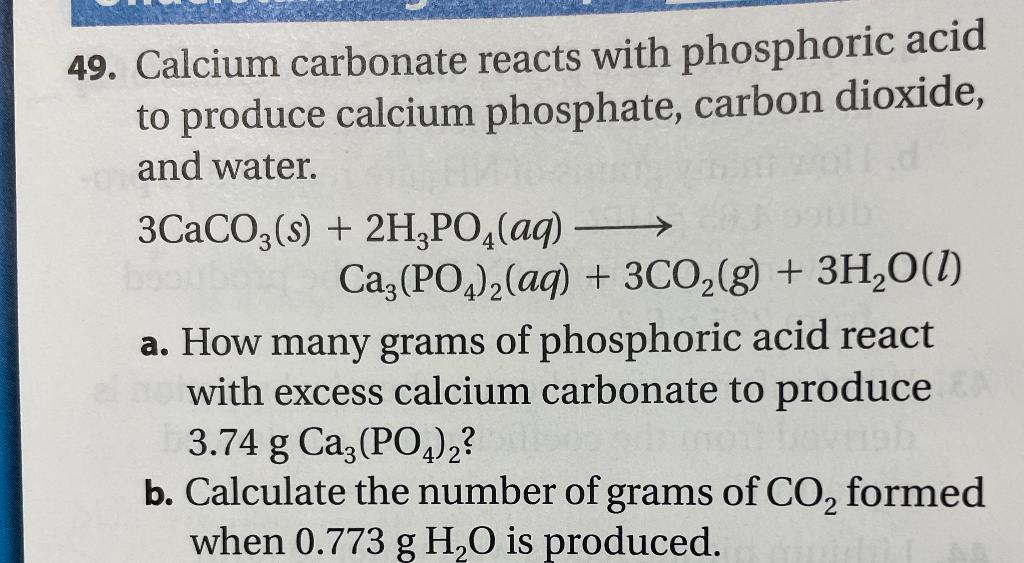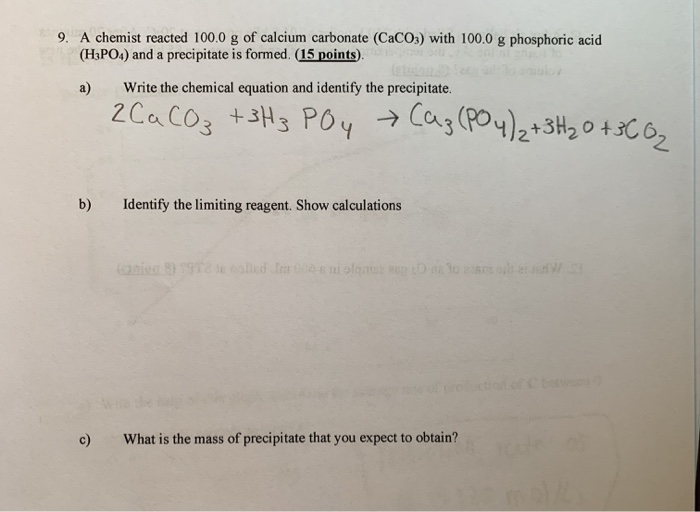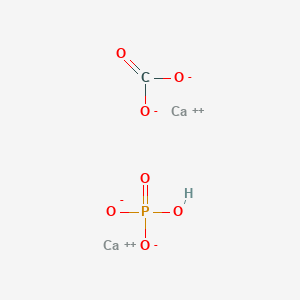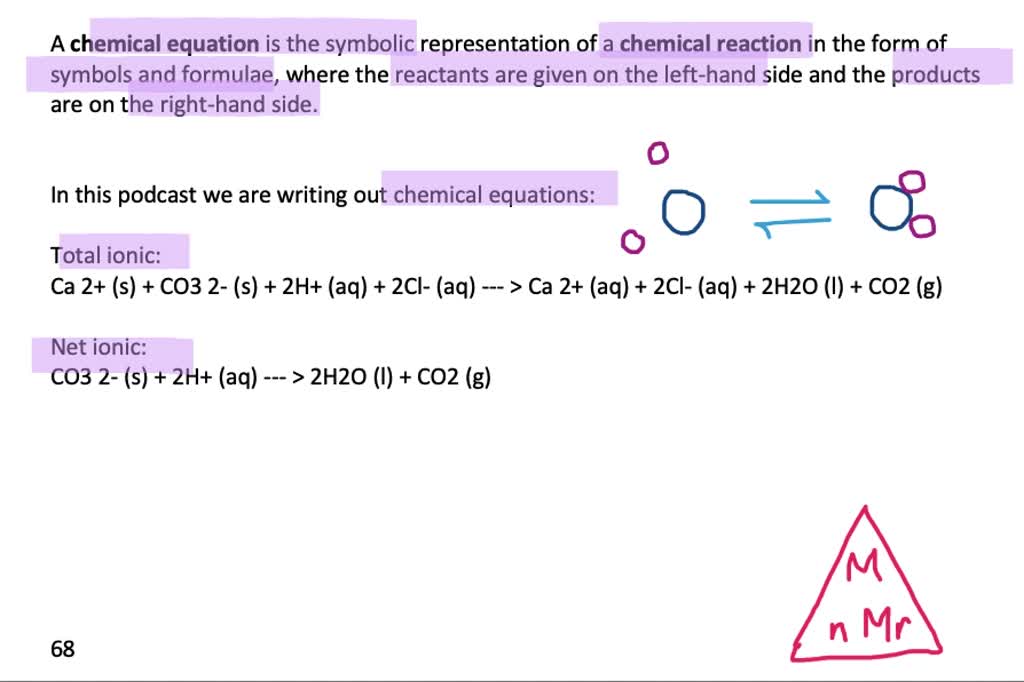
SOLVED: In the stomach, both the carbonate and phosphate ions react with the hydrochloric acid (HCl) in your stomach to become carbonic and phosphoric acid. When this happens, the calcium and chloride

Effect of pH and Phosphate on Calcium Carbonate Polymorphs Precipitated at near-Freezing Temperature | Crystal Growth & Design

The chemical analysis of the produced acidulated phosphoric acid solution. | Download Scientific Diagram

Factors controlling and influencing polymorphism, morphology and size of calcium carbonate synthesized through the carbonation route: A review - ScienceDirect

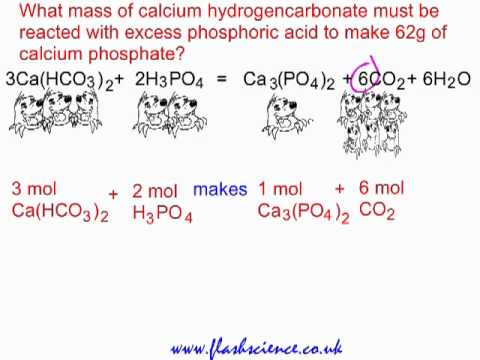
SOLVED: 13 (20 pts Potassium carbonate was neutralized with phosphoric acid as described below: KCO; (s) + 2 H;PO4 (aq) 2 K;PO4 (aq) + 3 COz (g) + H,O() (a) How many

ChemEngineering | Free Full-Text | Hydrothermal Synthesis of Biphasic Calcium Phosphate from Cuttlebone Assisted by the Biosurfactant L-rhamnose Monohydrate for Biomedical Materials

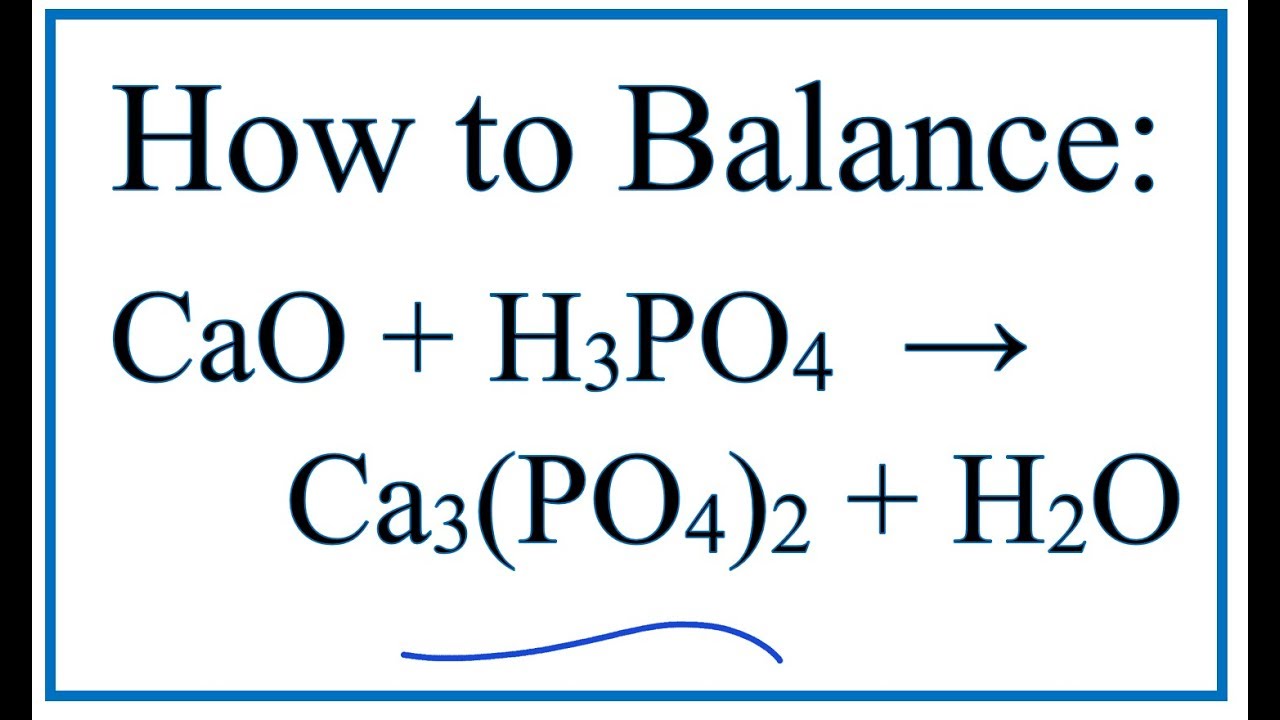
SOLVED:Write balanced chemical equations for each of the following processes: (a) Calcium phosphate reacts with sulfuric acid to produce calcium sulfate and phosphoric acid. (b) Calcium phosphate reacts with water containing dissolved