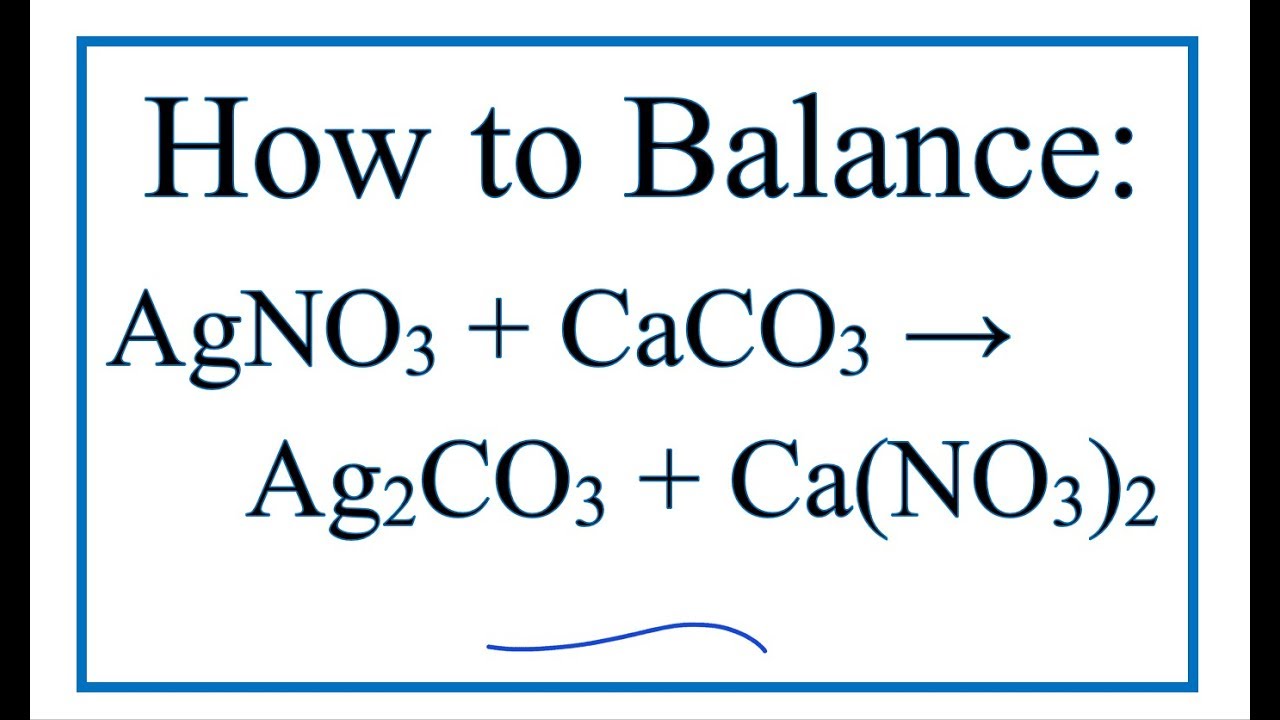
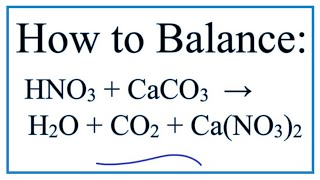
Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride

SOLVED: For the reaction Na2CO3+Ca(NO3)2⟶CaCO3+2NaNO3 how many grams of calcium nitrate, Ca(NO3)2, are needed to react completely with 55.5 g of sodium carbonate, Na2CO3?

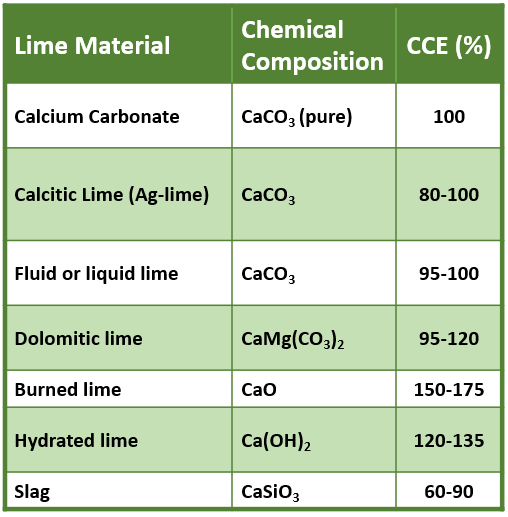
Limestone is held with tongs and heated on the flame directly. It crumbled to give white powder of calcium oxide. After cooling, when water is added to it a hissing sound is
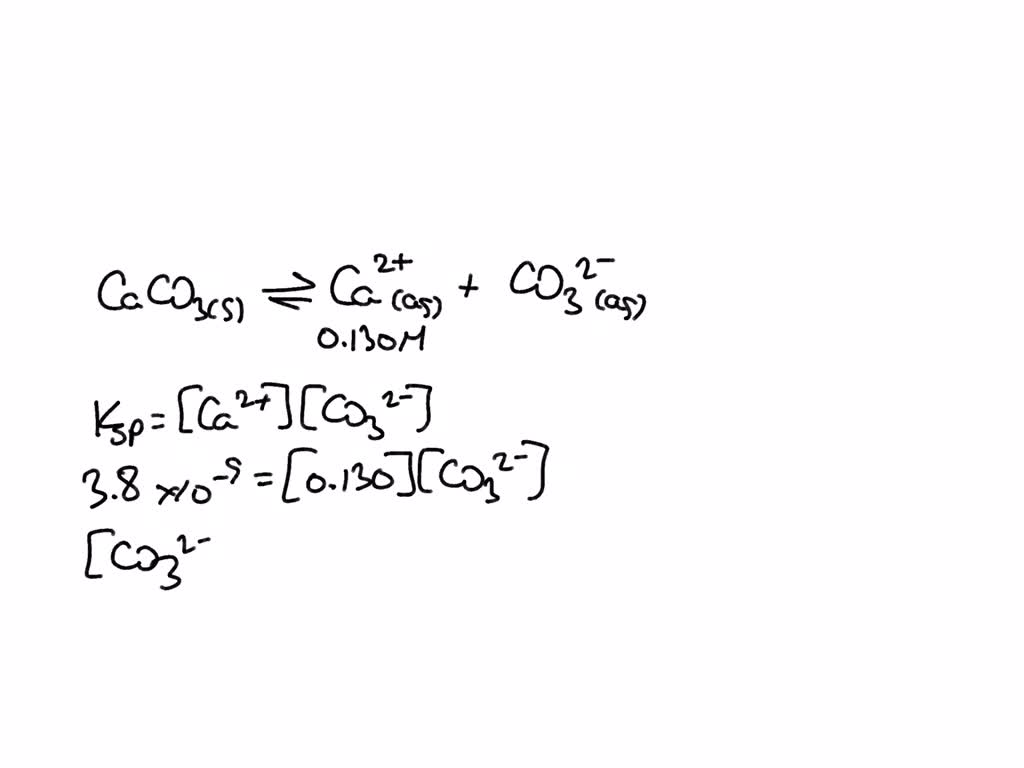
SOLVED: The molar solubility of calcium carbonate in a 0.130 M calcium nitrate solution is M. ksp of CaCO3 is 3.8 × 10-9

Toward the Mechanistic Understanding of the Additives' Role on Ammonium Nitrate Decomposition: Calcium Carbonate and Calcium Sulfate as Case Studies | ACS Omega

Factors controlling and influencing polymorphism, morphology and size of calcium carbonate synthesized through the carbonation route: A review - ScienceDirect

What happens when aqueous solutions of calcium chloride and of sodium carbonate are mixed? | Socratic