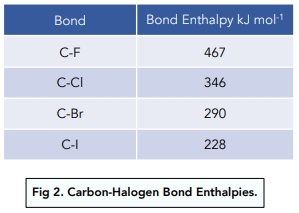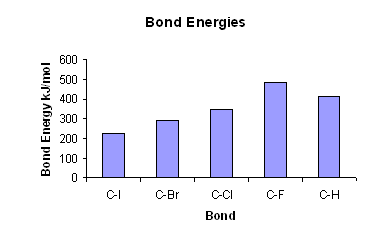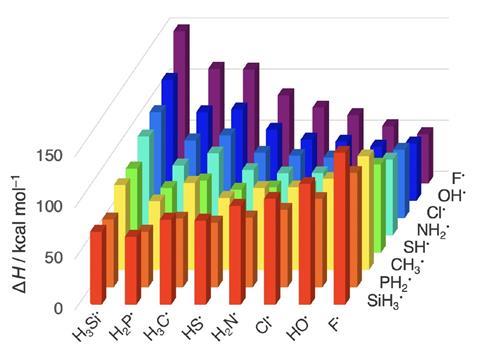
Textbook electronegativity model fails when it comes to carbon–halogen bond strengths | Research | Chemistry World

Reactivity and relative strength of C- Halogen bond in alkyl, allyl, benzyl, vinyl and aryl halides. - YouTube
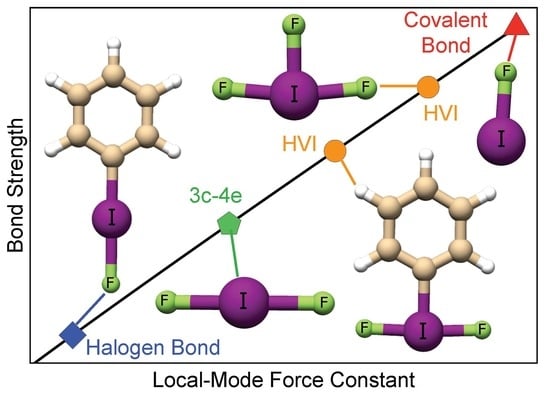
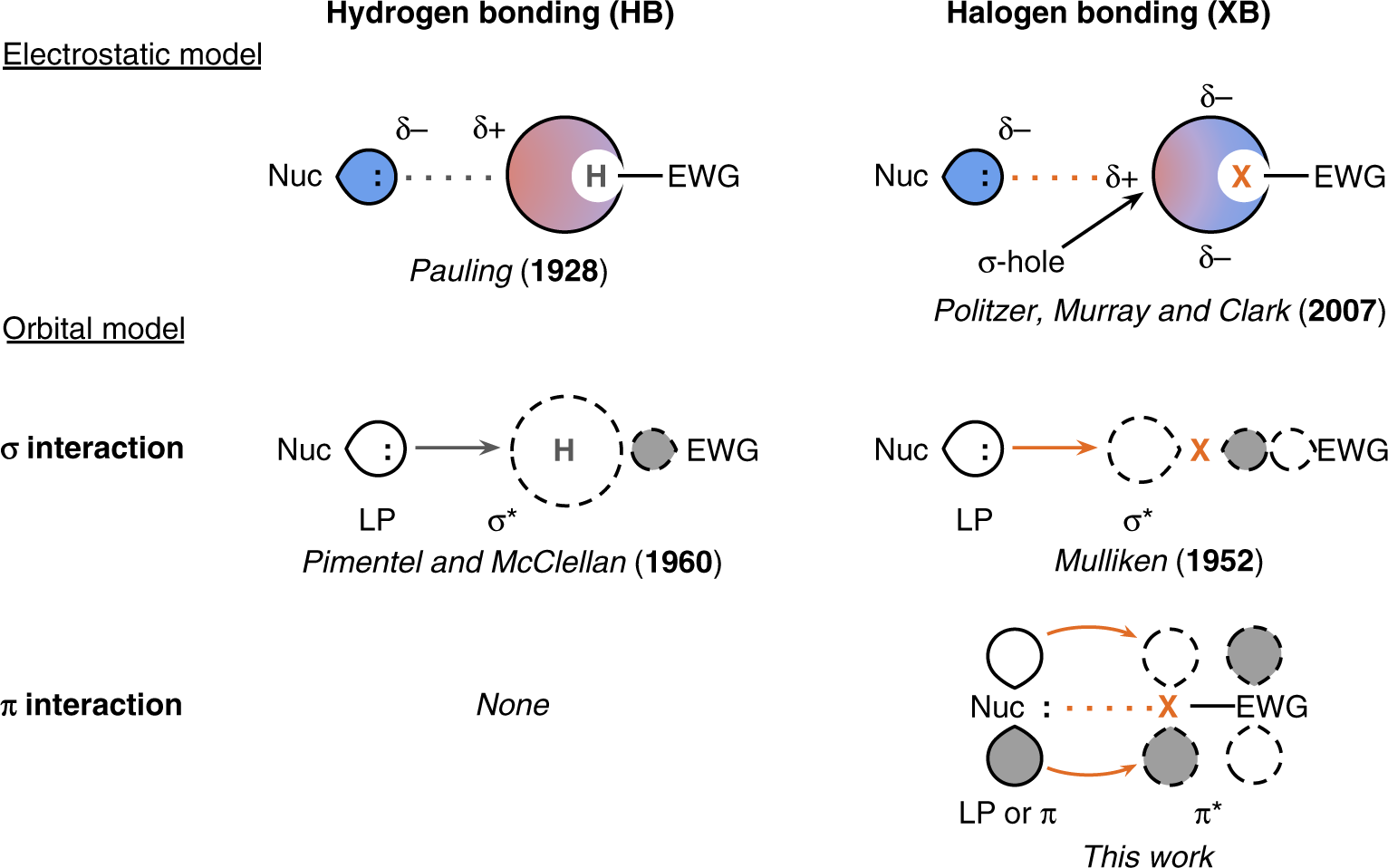
Inorganics | Free Full-Text | A Continuum from Halogen Bonds to Covalent Bonds: Where Do λ3 Iodanes Fit?
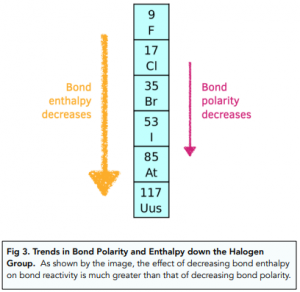
Why doesn't the bond polarity of a carbon halogen bond increase from Chlorine~Bromine~Iodine? - Quora

Palladium‐Catalyzed Activation of Carbon–Halogen Bonds: Electrostatics‐Controlled Reactivity - Moloto - 2022 - European Journal of Organic Chemistry - Wiley Online Library

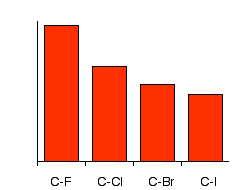
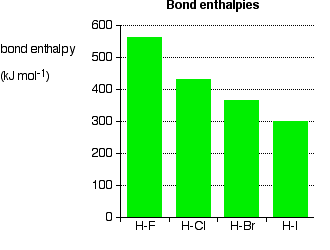
Physical Properties of Haloalkanes 6-1 The bond strength of C-X decreases as the size of X increases. A halogen uses a p orbital to overlap an sp 2 orbital. - ppt download