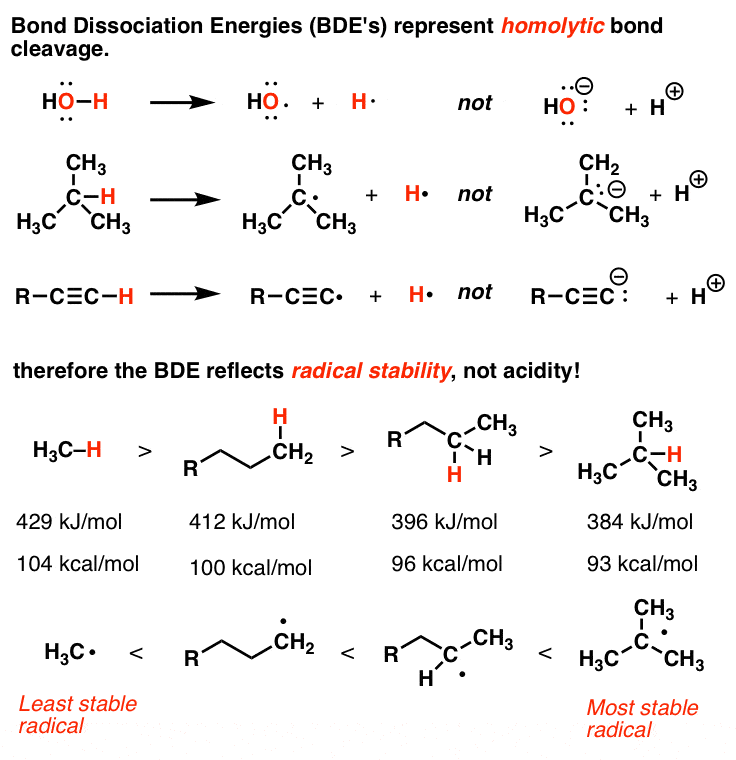
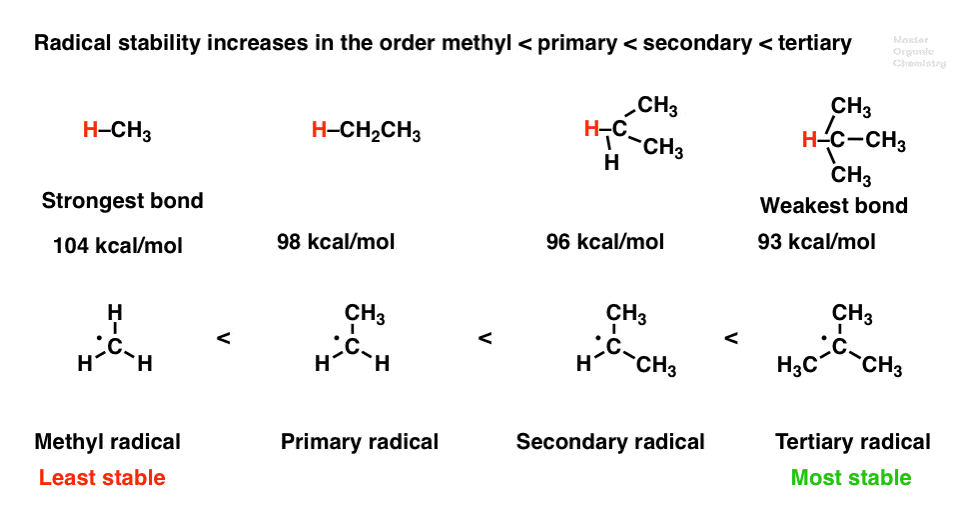
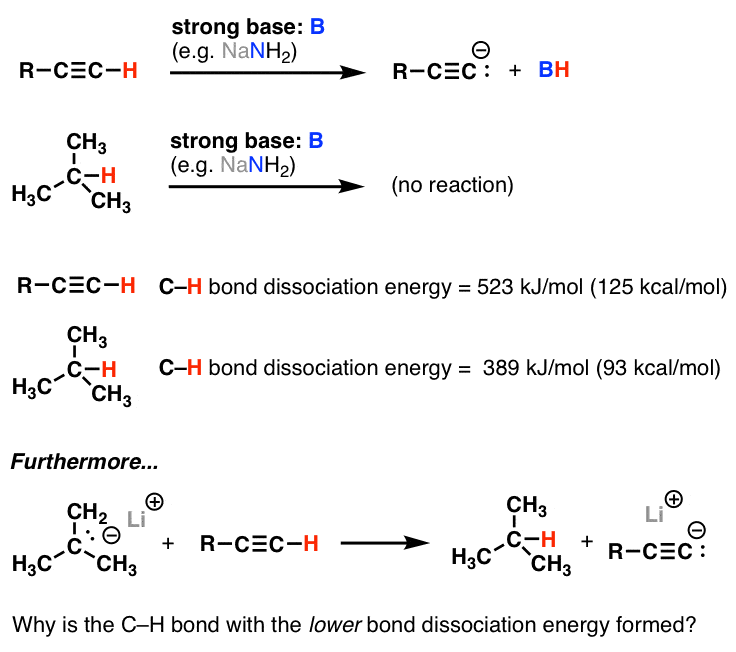
Rank the hydrogen according to their bond dissociation energy with carbon in descending order. | Homework.Study.com

Table 1 from Calculated carbon-hydrogen bond dissociation enthalpies for predicting oxidative susceptibility of drug substance molecules. | Semantic Scholar

Why bond energy of carbon hydrogen bond is in this order ethyne ethene ethane - Chemistry - Haloalkanes and Haloarenes - 13058453 | Meritnation.com

Cycloalkane and Cycloalkene C−H Bond Dissociation Energies | Journal of the American Chemical Society

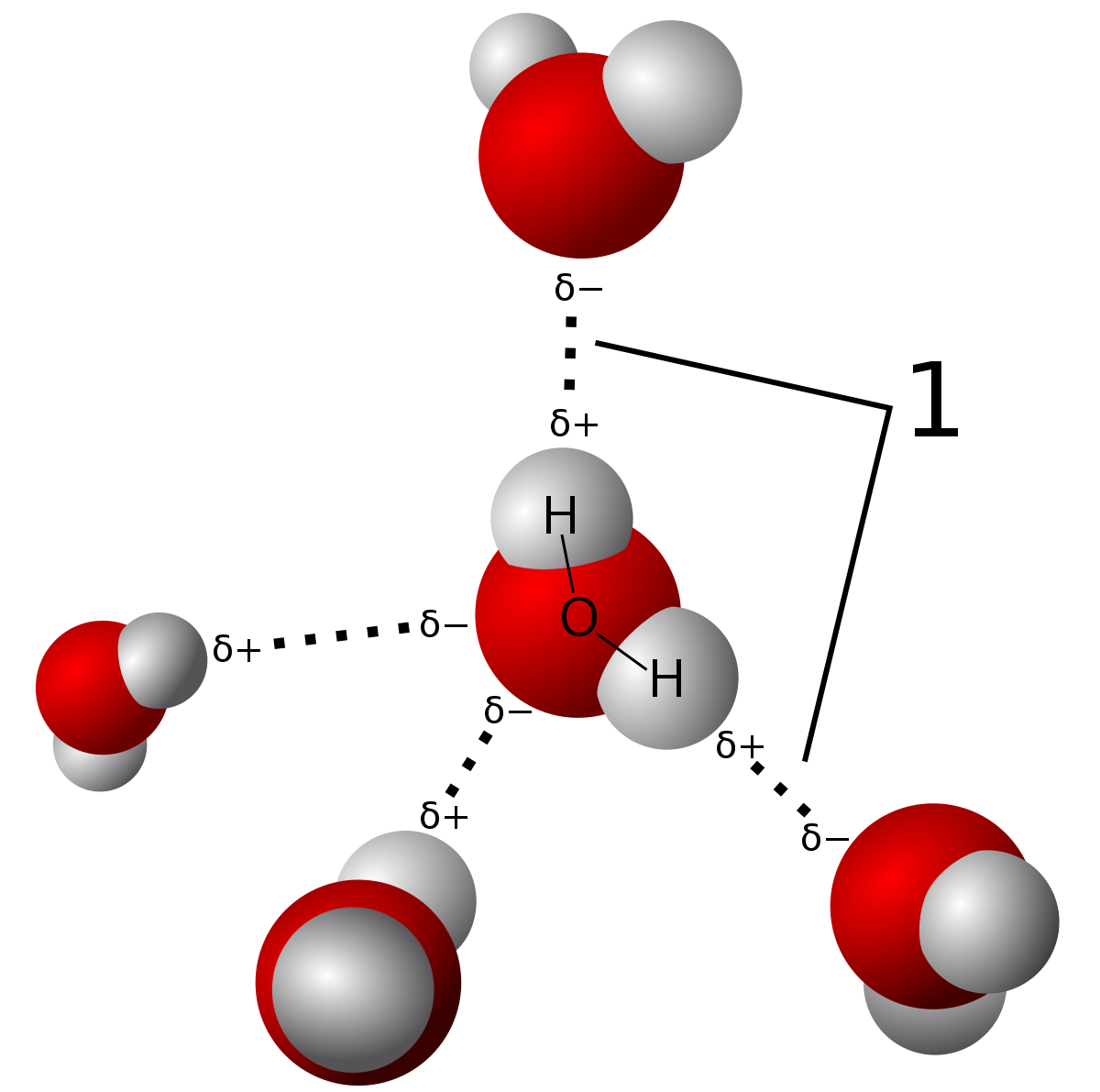
Hydrogen-Bond Network Promotes Water Splitting on the TiO2 Surface | Journal of the American Chemical Society

Cycloalkane and Cycloalkene C−H Bond Dissociation Energies | Journal of the American Chemical Society

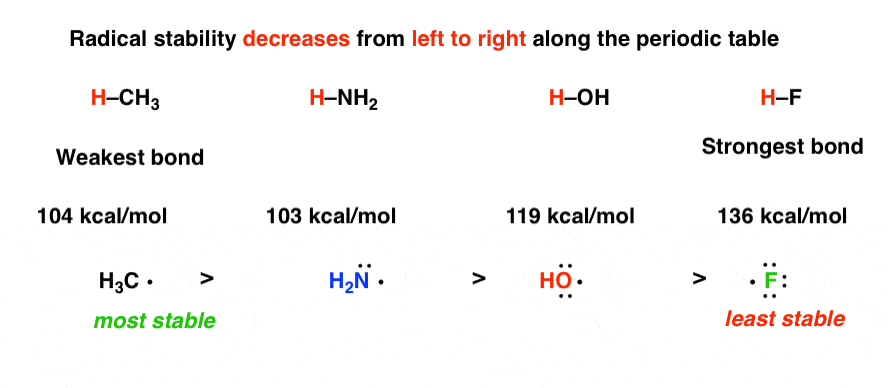
Why is breaking of C-H bond given more preference over C-C bond though the bond energy of the latter is less than the first one? – The Unconditional Guru

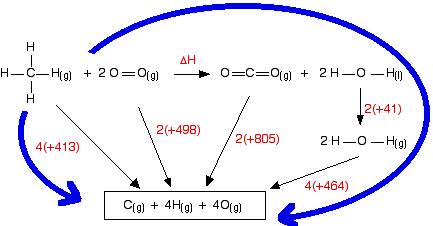
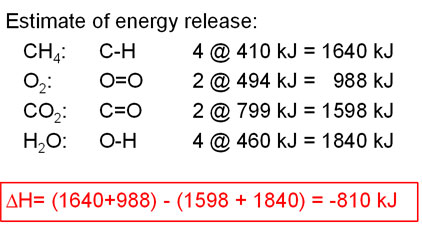
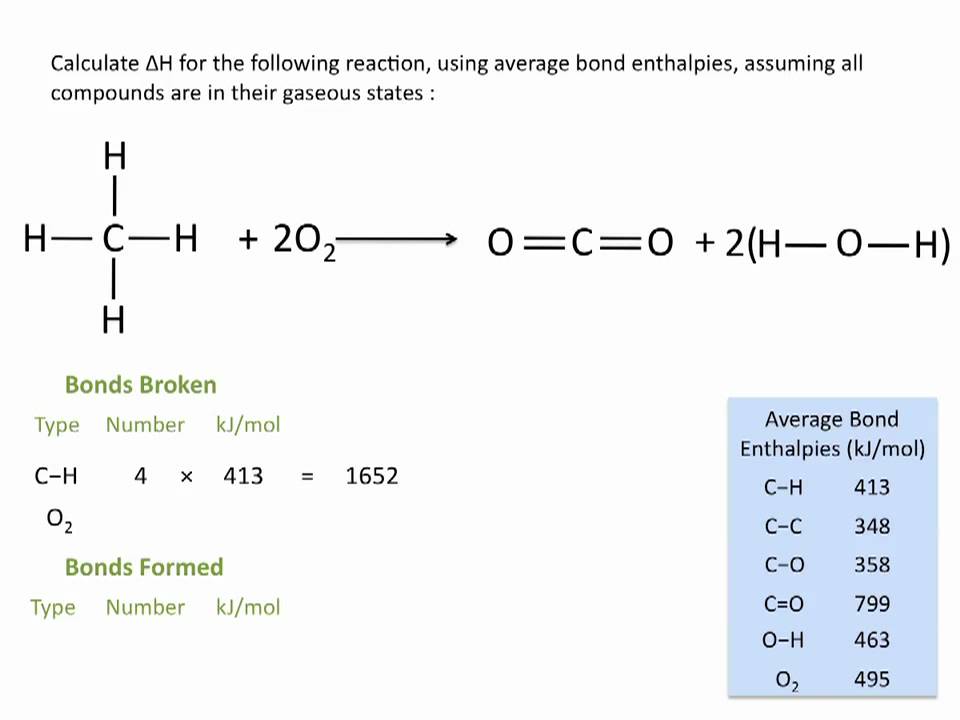
Chemical reactions are invariably associated with the transfer of energy either in the form of heat or light. In the laboratory, heat changes in physical and chemical processes are measured with an