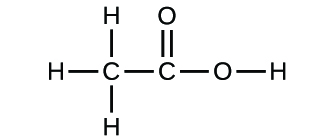
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

An organic compound contains 20 % carbon, 6.7 % hydrogen, and 46.67 % nitrogen. Its molecular weight was found to be 60 . Find the molecular formula of the compound.
12. An organic compound of carbon, hydrogen andnitrogen contains these elements having masspercentage 66.67

The empirical formula weight of a compound containing carbon and hydrogen is 13 . The molecule of the compound is 39 times heavier than a molecule of hydrogen. The molecular formula of
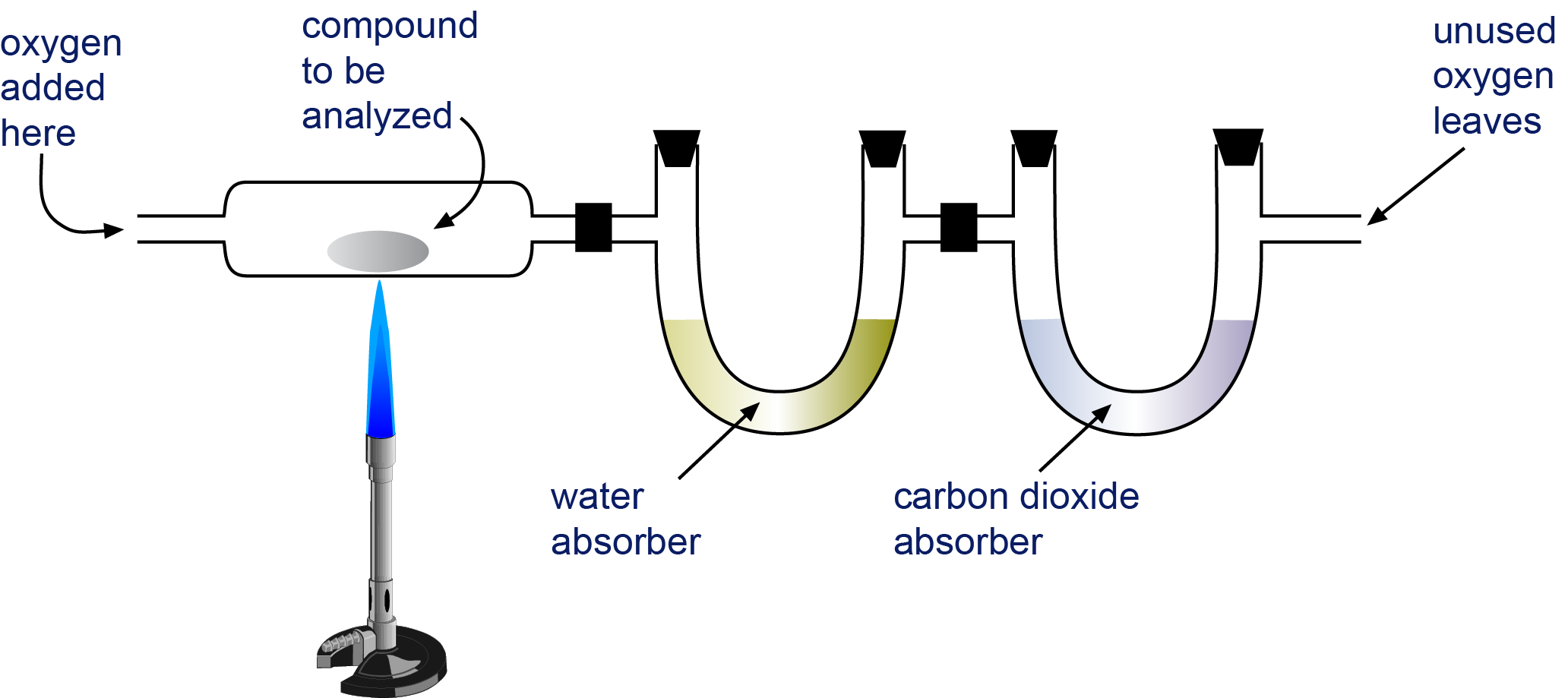
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

Question Video: Determining the Number of Carbon Atoms in an Alkene Given the Number of Hydrogen Atoms | Nagwa