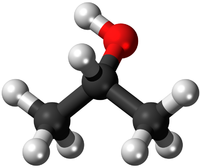
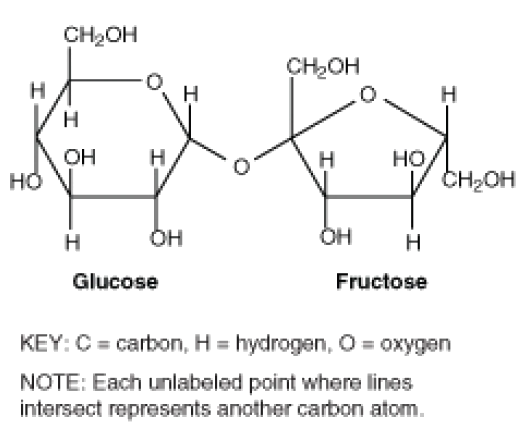
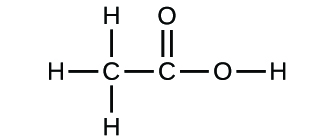
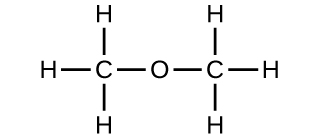
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

An organic compound contains carbon, hydrogen and oxygen. Its elemental analysis gave`C,38.71% - YouTube
.png)
an organic compound containing carbon ,hydrogen and oxygen gave the following composition : C- 54 55% ,H 9 09% and - Chemistry - - 9449851 | Meritnation.com

A compound containing only carbon, hydrogen and oxygen was analyzed and found to contain 3.25 % hydrogen and 19.36 % carbon. What is the empricial formula of the compound? | Homework.Study.com

Carbon, hydrogen, oxygen, nitrogen, chlorine, and tin atoms are shown... | Download Scientific Diagram

An organic compound contains carbon, hydrogen and oxygen. If the ratio percentage of C and - YouTube