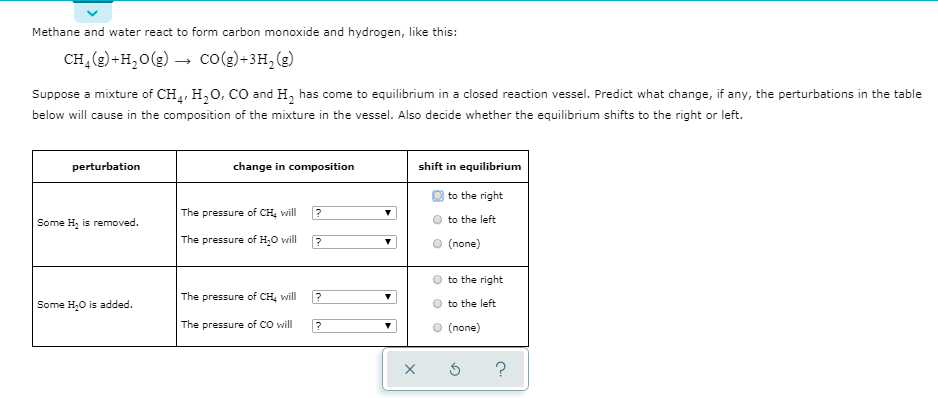
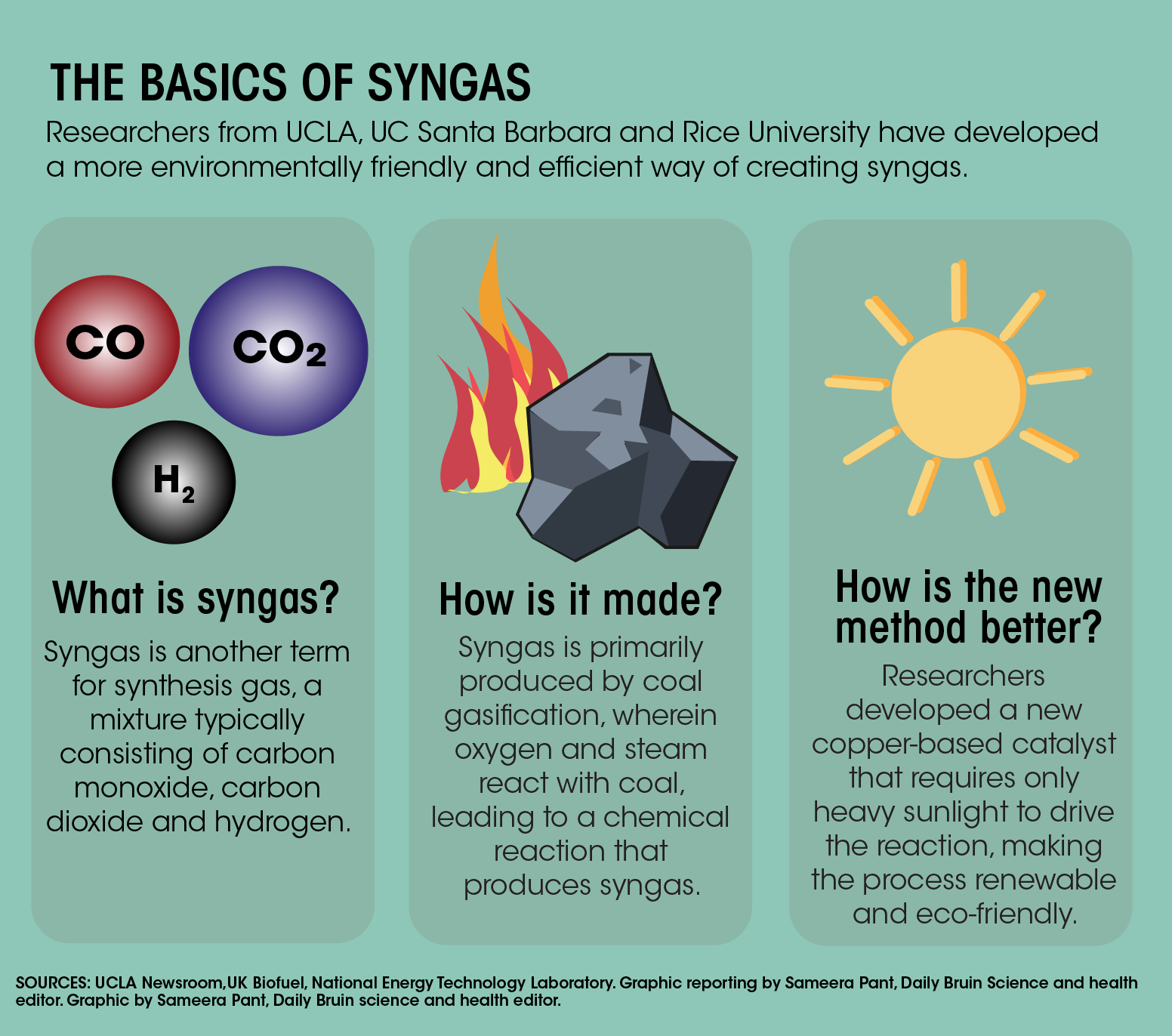
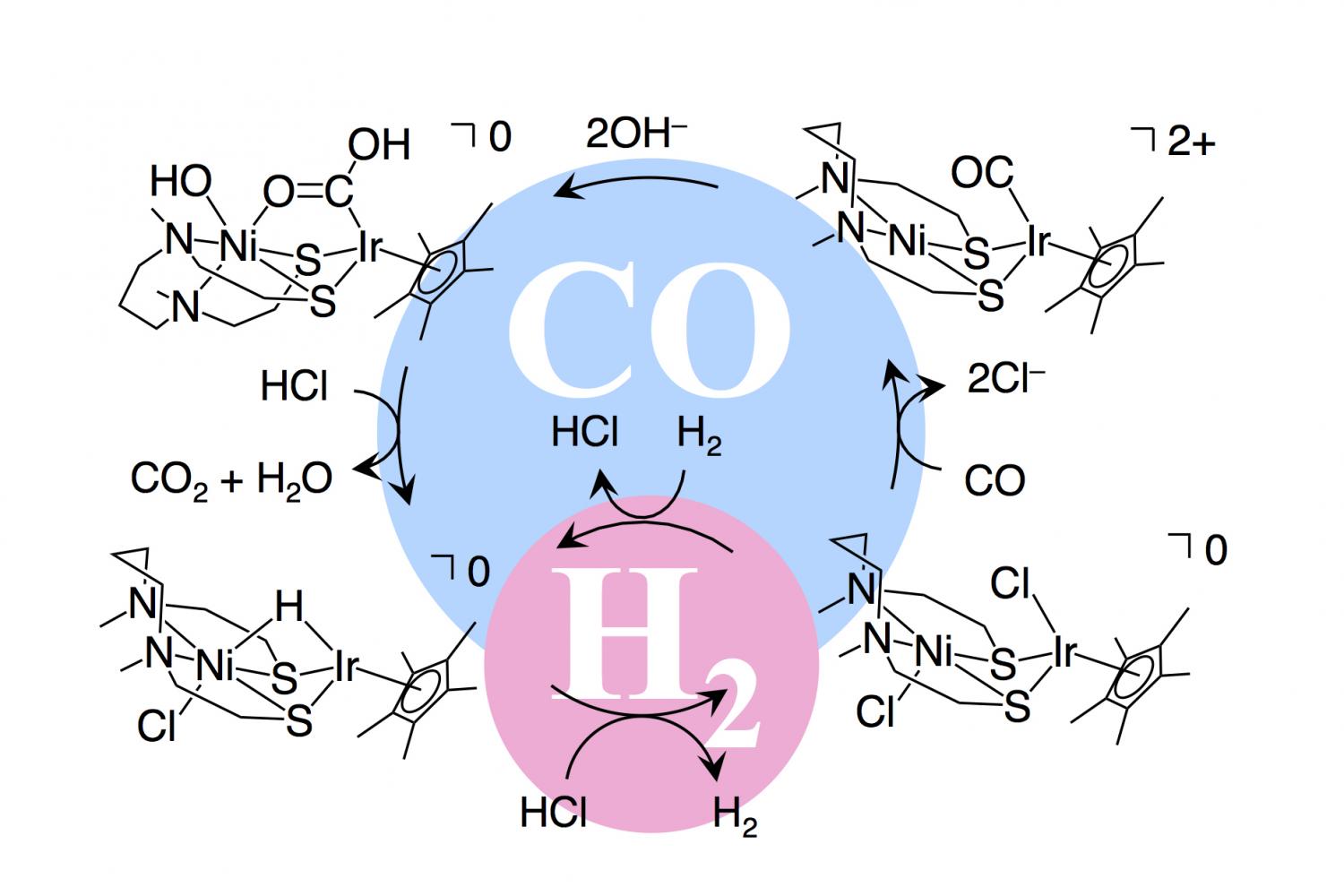
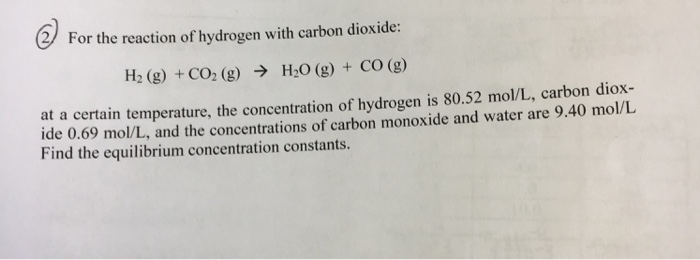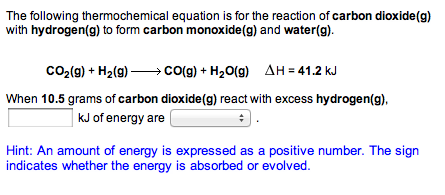Propene reacts with carbon monoxide and hydrogen in presence of cobalt carbonyl catalyst at high - YouTube

SOLVED: 41 Methanol is formed by reacting carbon monoxide with hydrogen. (This might be an on-board generation of hydrogen for the hydrogen economy) With the reaction CO + 2Hz CH;OH (4.17) the

Carbon monoxide reacts with hydrogen under certain conditions to from methanol `(CH_(3)OH)` - YouTube
![SOLVED: Methanol is formed from carbon monoxide and hyarogen gas according to the following balanced equation: CO(g) 2Hz (g) CH; OHcg) a) [5 pts] Using the values provided in the table; calculate SOLVED: Methanol is formed from carbon monoxide and hyarogen gas according to the following balanced equation: CO(g) 2Hz (g) CH; OHcg) a) [5 pts] Using the values provided in the table; calculate](https://cdn.numerade.com/ask_images/02c7b6f2431a40e6bdae48e298e6cc4a.jpg)
SOLVED: Methanol is formed from carbon monoxide and hyarogen gas according to the following balanced equation: CO(g) 2Hz (g) CH; OHcg) a) [5 pts] Using the values provided in the table; calculate
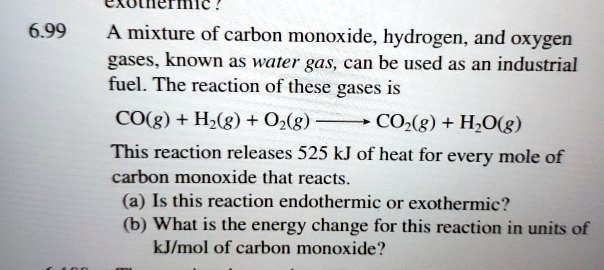
SOLVED: CAOICMIC 6.99 A mixture of carbon monoxide, hydrogen, and oxygen gases; known as water gas; can be used as an industrial fuel. The reaction of these gases CO(g) + Hz(g) +

Reduction of carbon dioxide with hydrogen on a CuO–ZnO mixed catalyst under supercritical conditions - ScienceDirect

hydrocarbons - Why carbon monoxide reacts with hydrogen to give different products in the presence of different catalysts - Chemistry Stack Exchange

Natural gas consists primarily of methane, CH 4.It is used in a process called stream reforming to prepare a gaseous mixture of carbon monoxide and hydrogen. - ppt download




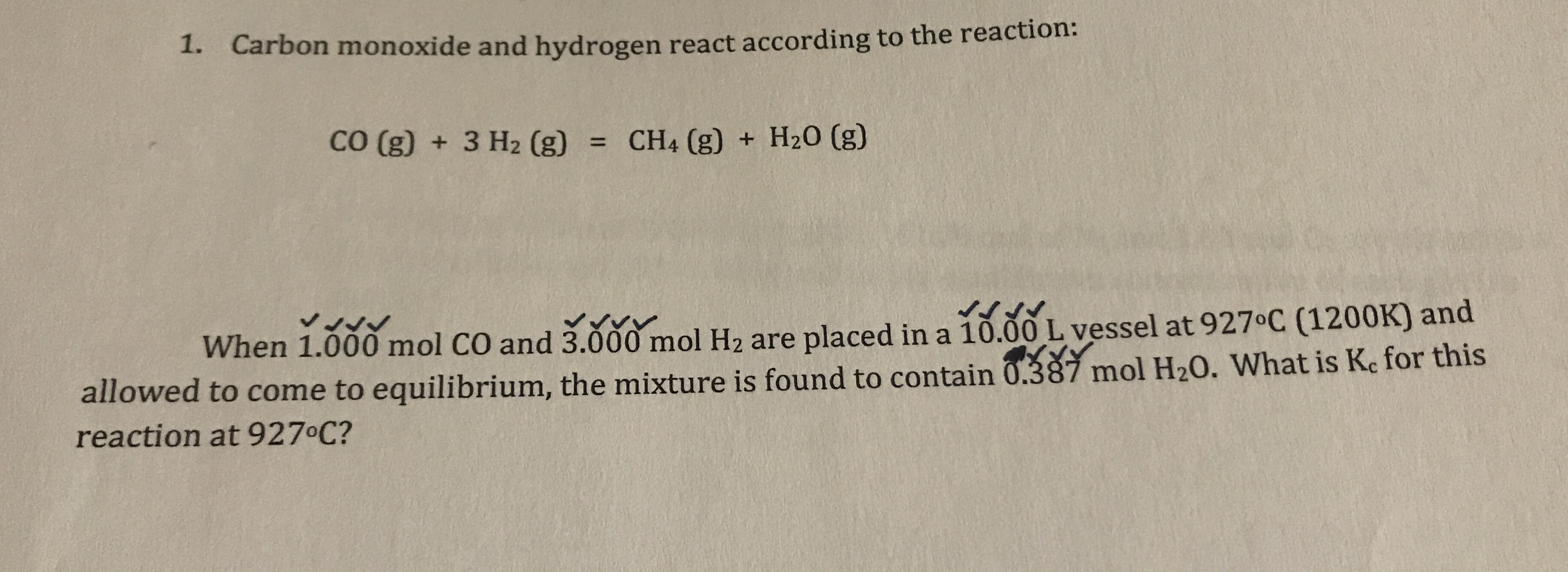
.jpg)