A) At 10 atm, what are the melting and boiling points of Carbon Dioxide? B) At -20^(\circ)C, at what pressure does Carbon Dioxide boil and freeze? C) At 20^(\circ)C and 10 atm,

Carbon monoxide Authors: Dr. Bajnóczy Gábor Kiss Bernadett BUDAPEST UNIVERSITY OF TECHNOLOGY AND ECONOMICS DEPARTMENT OF CHEMICAL AND ENVIRONMENTAL PROCESS. - ppt download
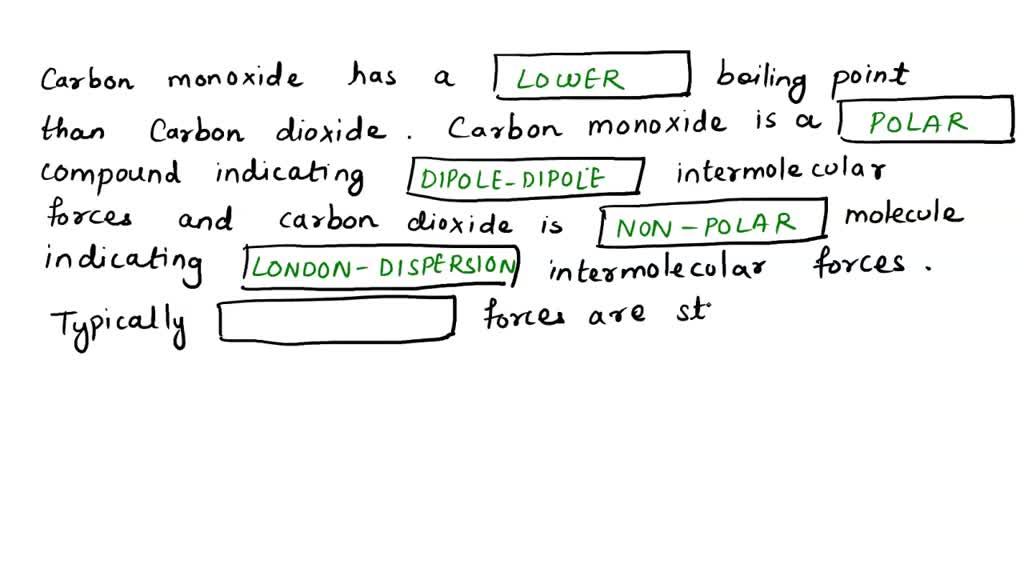
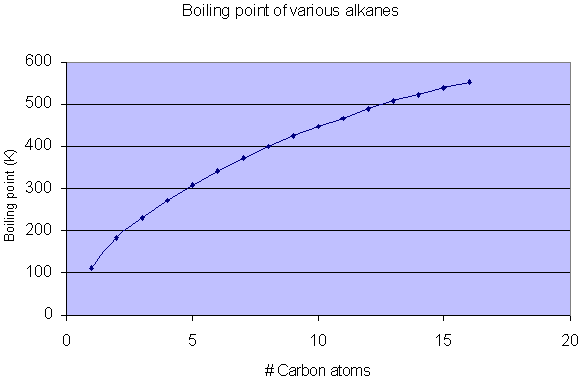
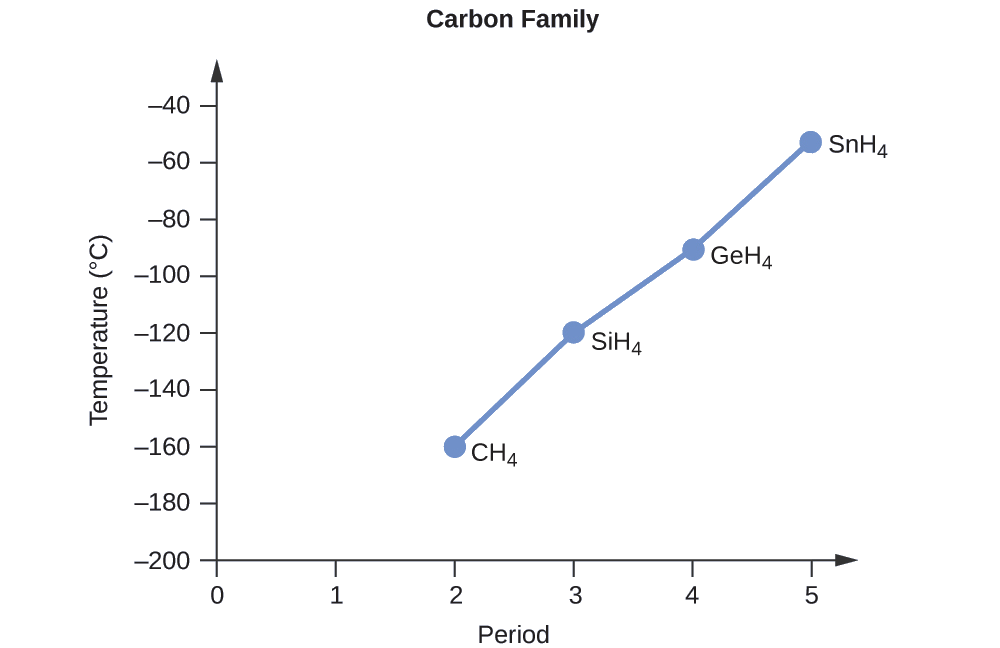
SOLVED: Look up the boiling points of carbon monoxide and carbon dioxide. Which intermolecular force would you cite to account for the difference? Explain.



![Boiling temperature vs. pressure for CO2 [6] | Download Scientific Diagram Boiling temperature vs. pressure for CO2 [6] | Download Scientific Diagram](https://www.researchgate.net/publication/266441851/figure/fig4/AS:456125523992580@1485760207188/Boiling-temperature-vs-pressure-for-CO2-6.png)

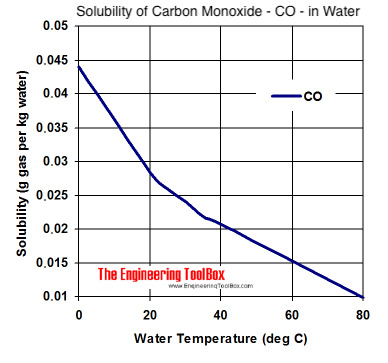
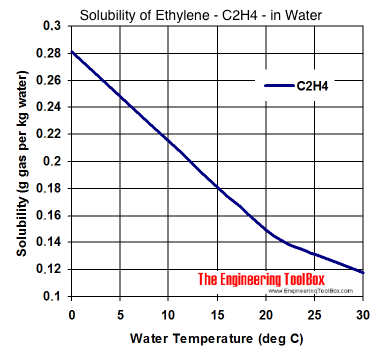
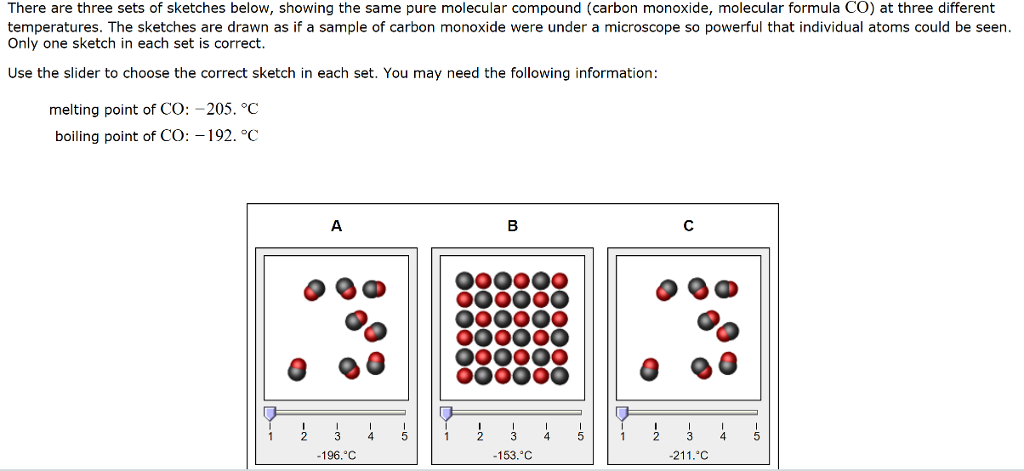

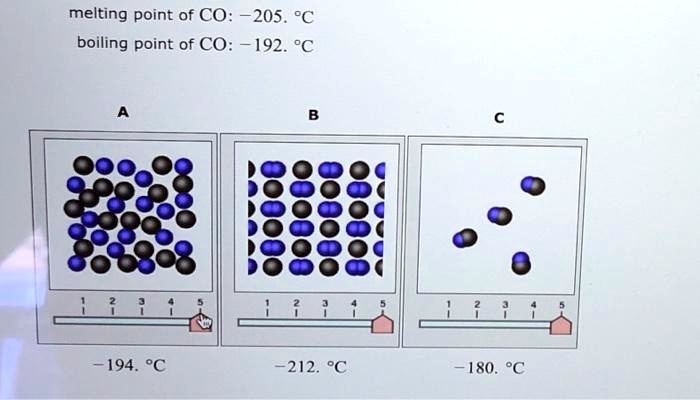


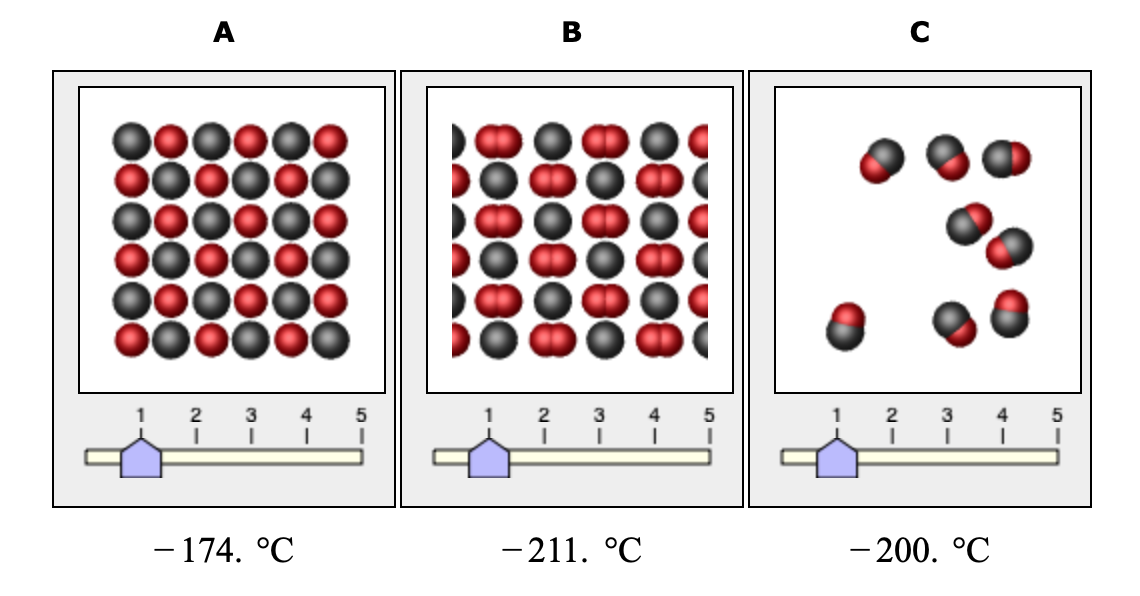


![Physical Properties of Carbon monoxide[II] | Download Scientific Diagram Physical Properties of Carbon monoxide[II] | Download Scientific Diagram](https://www.researchgate.net/publication/338537906/figure/tbl1/AS:857009956081669@1581338504914/Physical-Properties-of-Carbon-monoxideII.png)