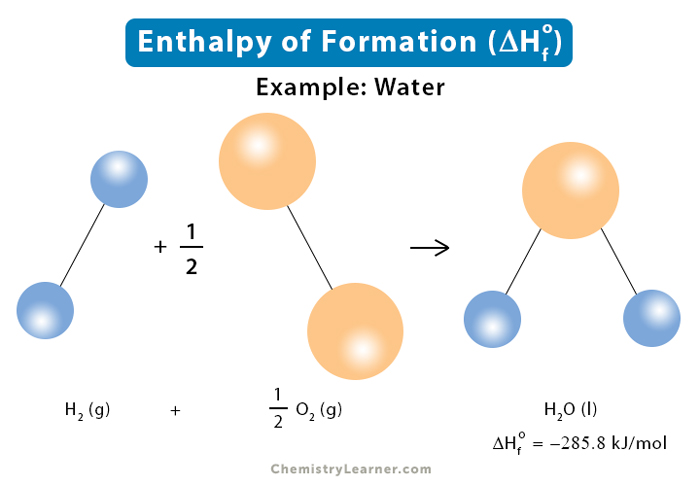
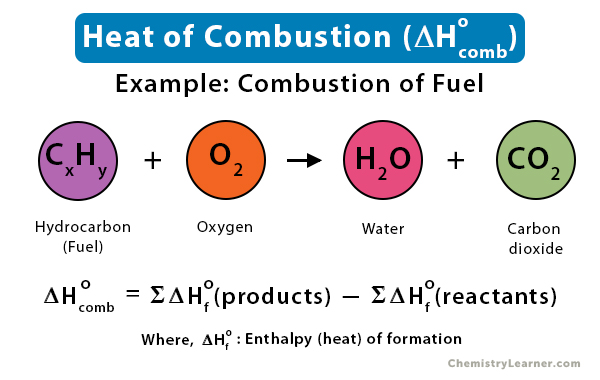
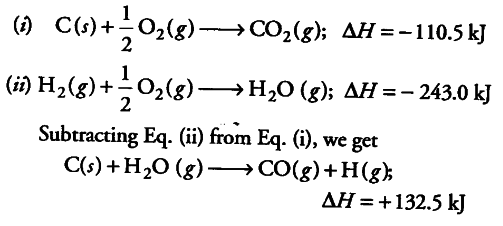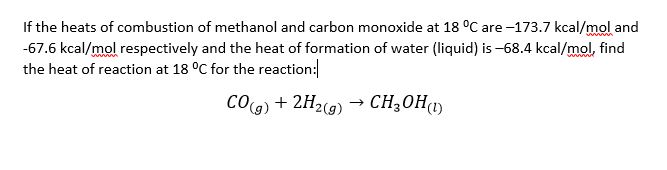Given standard enthalpy of formation of CO ( - 110 KJ mol^-1 ) and C O2 ( - 394 KJ mol^-1 ). The heat of combustion when one mole of graphite burns is:

SOLVED: An important industrial route t0 extremely pure acetic acid is the reaction of methanol with carbon monoxide: Use bond energies t0 calculate the heat of reaction Average Bond Energies Bond Bond

Calculate the enthalpy of formation of carbon monoxide (CO) from the following data: (i) C (s) + O2 (g) → - Brainly.in

The heat of formation of CO(g) and CO2(g) are Δ H = - 110 and Δ H = - 393kJmmol^-1 respectively. What is the heat of reaction (Δ H) (in kJ mol^-1 )
The enthalpies of combustion of carbon and carbon monoxide are -393.5 and –283kJ mol^–1 respectively. - Sarthaks eConnect | Largest Online Education Community
Heat of combustion of carbon monoxide is 283.5 kJ/mole the heat released when 55g of carbon dioxide formed from carbon monoxide