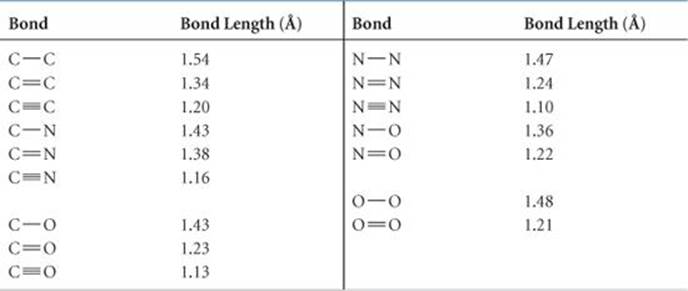
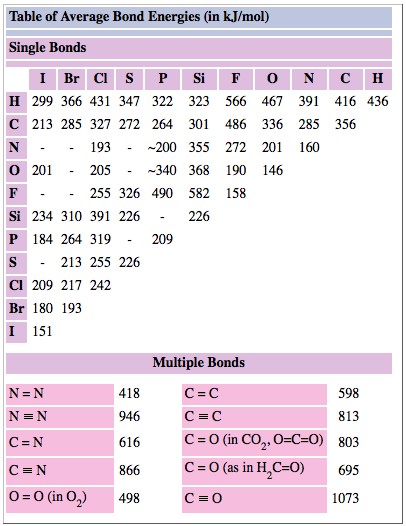
![PDF] Bond Length - Bond Valence Relationships for Carbon - Carbon and Carbon - Oxygen Bonds | Semantic Scholar PDF] Bond Length - Bond Valence Relationships for Carbon - Carbon and Carbon - Oxygen Bonds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/229ac8099c4a475d47a8b557fd14326f02189409/4-Table1-1.png)
PDF] Bond Length - Bond Valence Relationships for Carbon - Carbon and Carbon - Oxygen Bonds | Semantic Scholar

Give reasons for the following : 'Carbon-oxygen bond lengths in formic acid aer `1.23 Å` and `1.36 - YouTube

the correct arrangement of the species in the decreasing order of bond length between carbon and oxygen - Brainly.in

SOLVED:Order the following species with respect to carbon–oxygen bond length (longest to shortest). CO, CO2, CO3^2-, CH3 OH What is the order from the weakest to the strongest carbon–oxygen bond? (CH3 OH
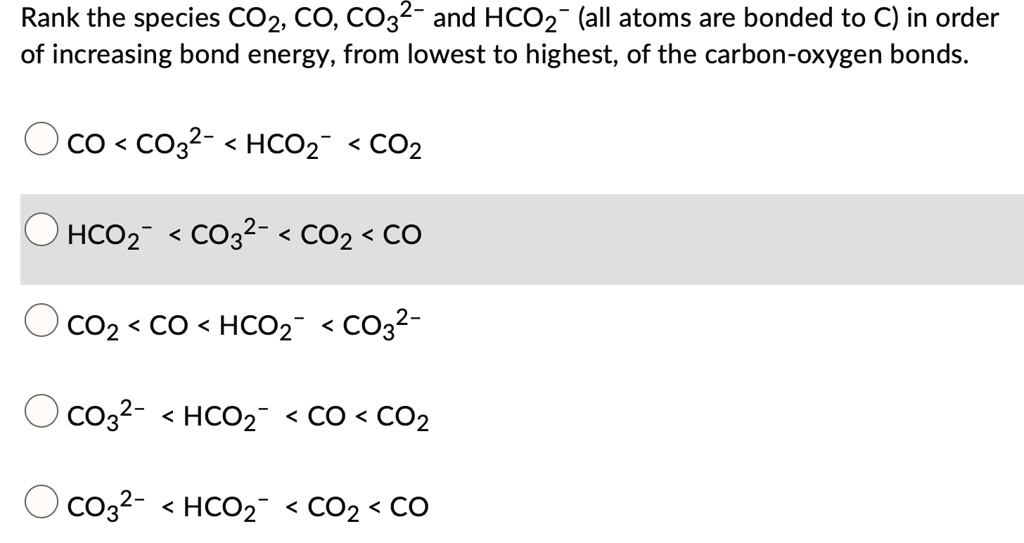
SOLVED: Rank the species CO2, CO, CO32- and HCO2" (all atoms are bonded to C) in order of increasing bond energy, from lowest to highest; of the carbon -oxygen bonds: CO CO32 -

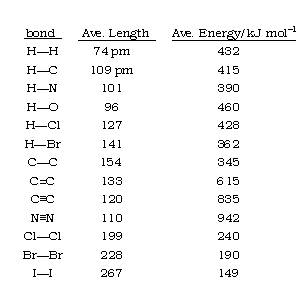



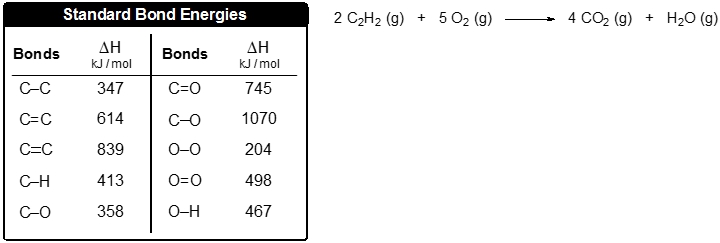
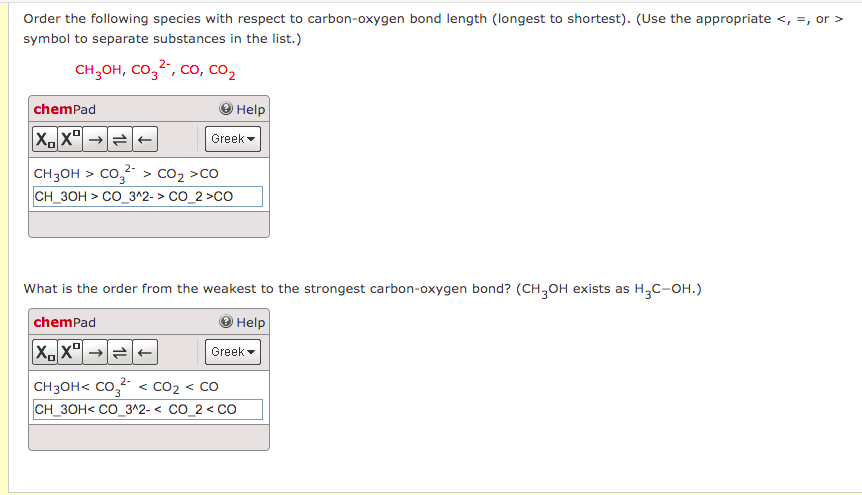
![PDF] A Bond Length – Bond Valence Relationship for Carbon – Nitrogen Bonds | Semantic Scholar PDF] A Bond Length – Bond Valence Relationship for Carbon – Nitrogen Bonds | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/568aa242179ebd3bef9e04c0584a1123c3e2a6c3/3-Figure1-1.png)