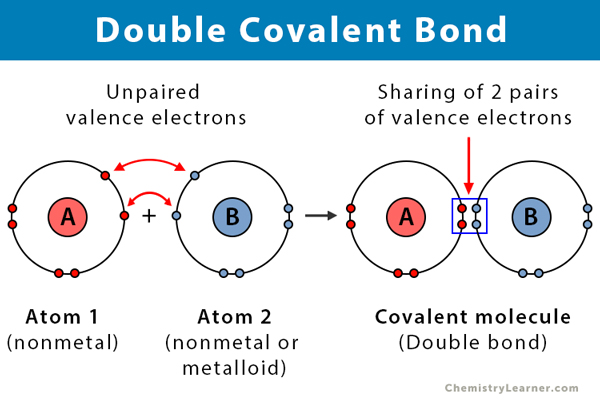
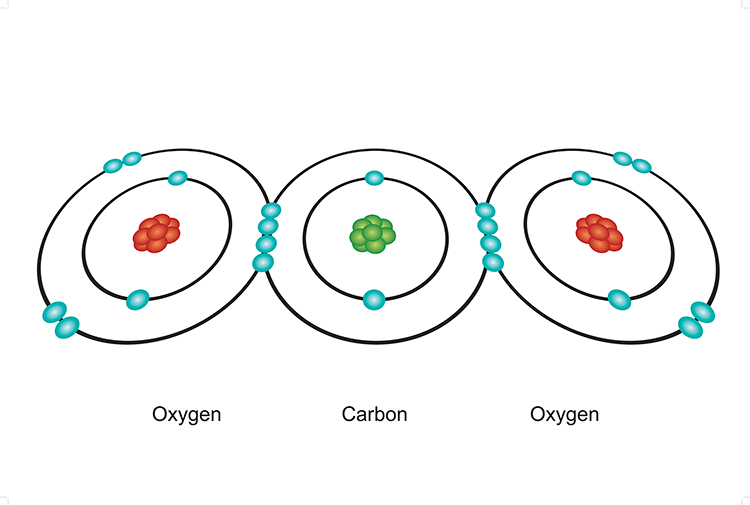
carbon dioxide molecule CO2 Lewis dot & cross electronic diagram covalent bonds ball & stick space filling 3D models boiling point melting point Doc Brown's chemistry revision notes
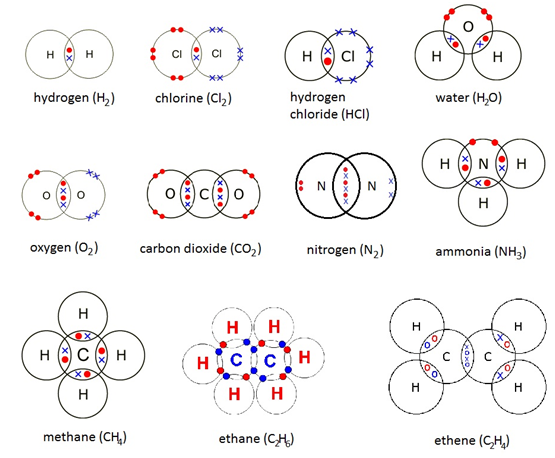
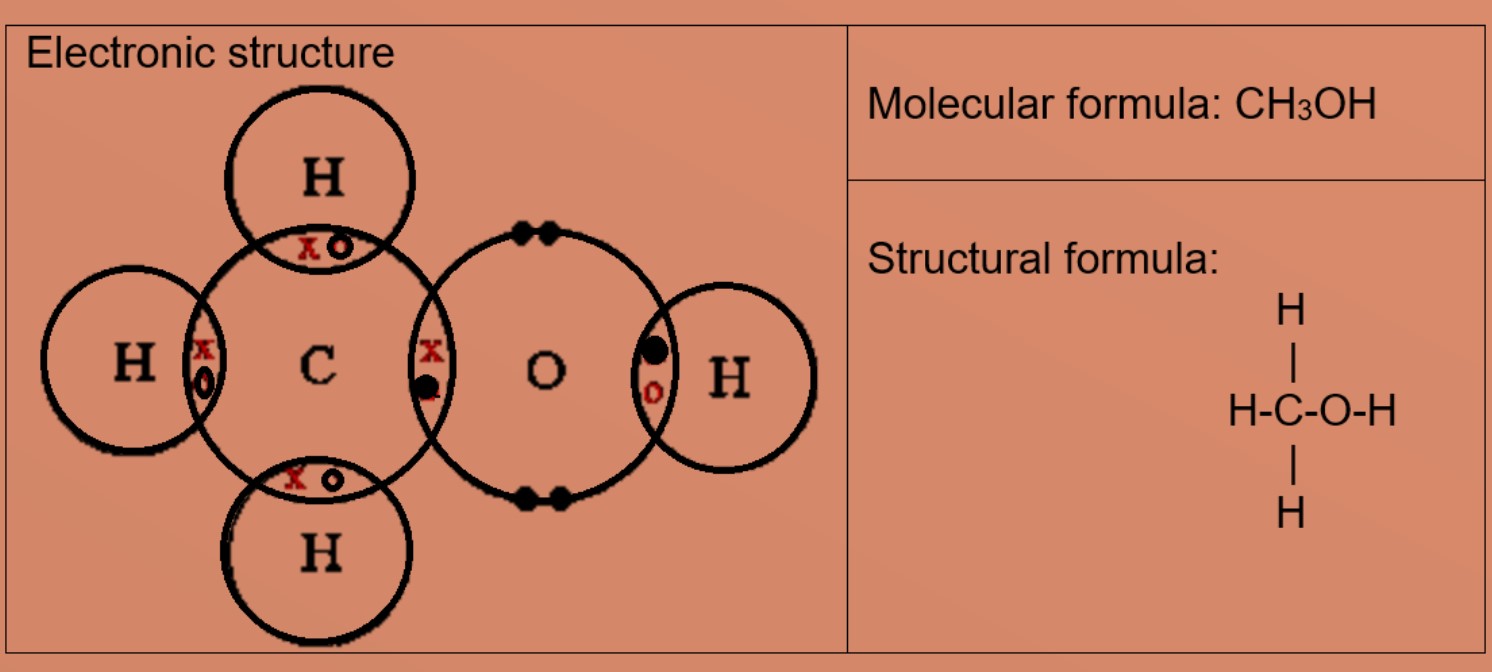
1:46 understand how to use dot-and-cross diagrams to represent covalent bonds in: diatomic molecules, including hydrogen, oxygen, nitrogen, halogens and hydrogen halides, inorganic molecules including water, ammonia and carbon dioxide, organic molecules
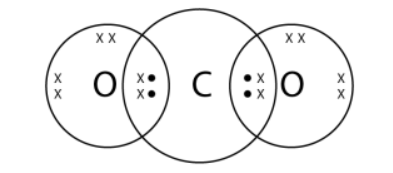
Draw the electron-dot structures of the following compounds and state the type of bonding in each case: - Noon Academy

Multimedia: Represent Bonding with Lewis Dot Diagrams | Chapter 4, Lesson 6 | Middle School Chemistry

Which of the following statements is true regarding a covalent bond in CO? a. Valence electrons are transferred to the oxygen atom. b. The bond length is less than the sum of




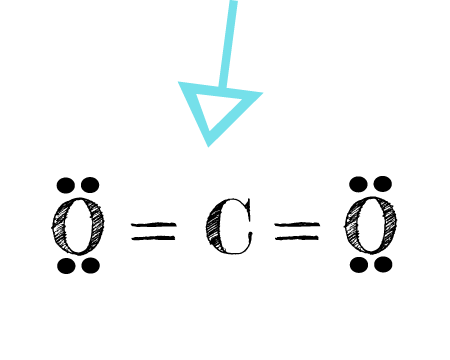

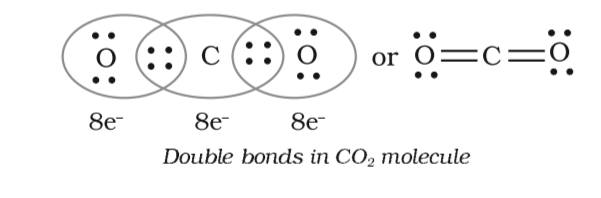
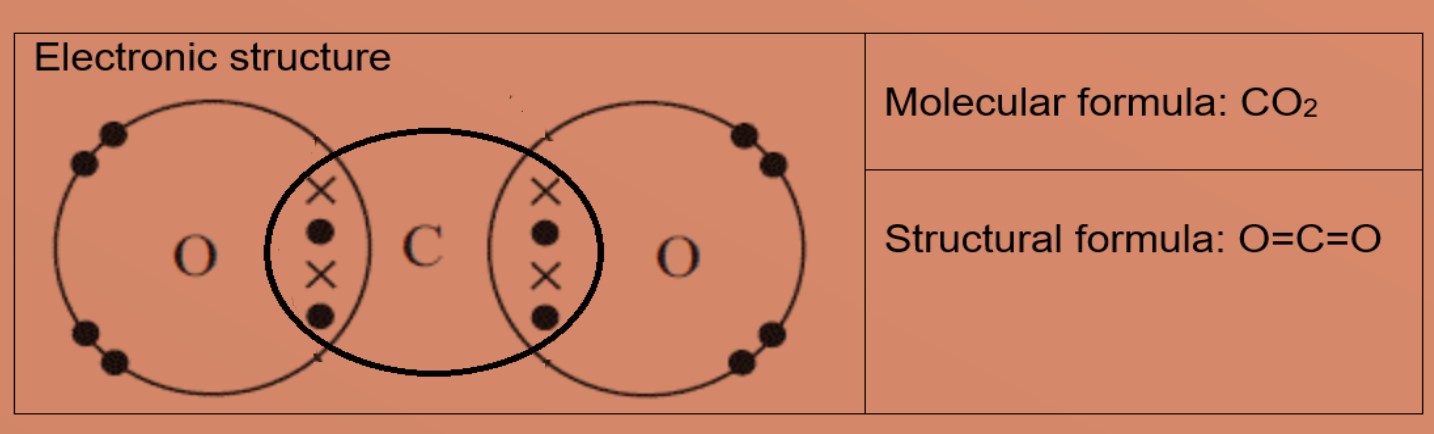
![Class 10] Electron dot structure of carbon dioxide with formula CO2 Class 10] Electron dot structure of carbon dioxide with formula CO2](https://d1avenlh0i1xmr.cloudfront.net/c50548eb-9014-4a63-9802-3fb5a77c60a2/covalent-bonding-in-co2---teachoo.jpg)

:max_bytes(150000):strip_icc()/nitrogen-molecule-122373927-57d6b2463df78c58336b37f5.jpg)