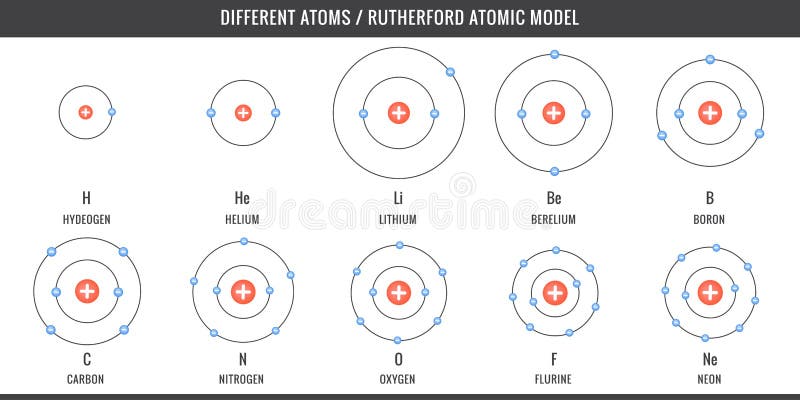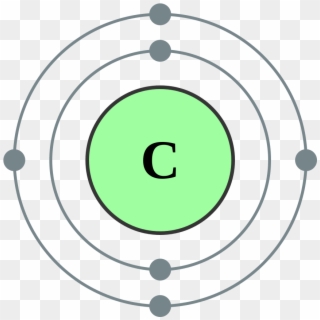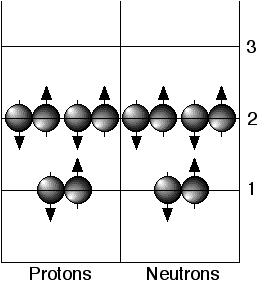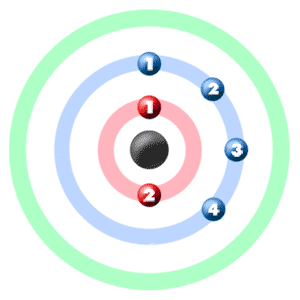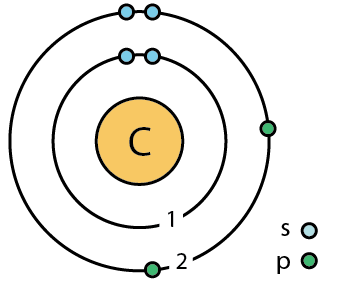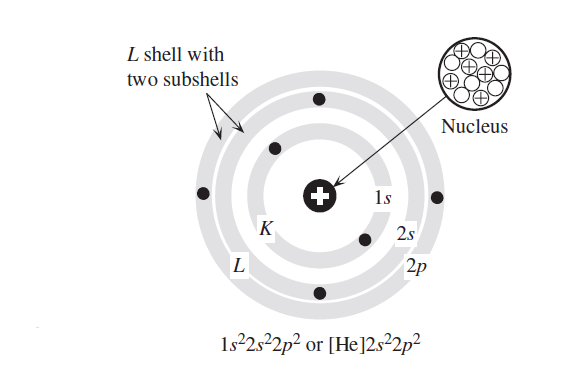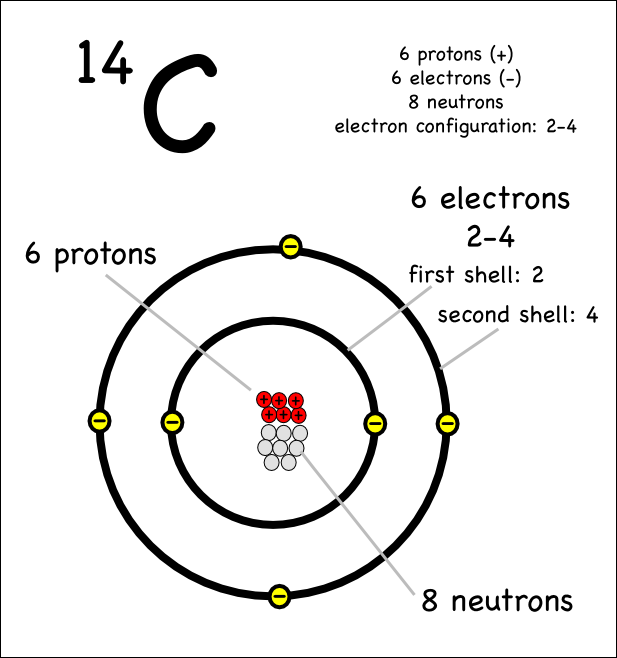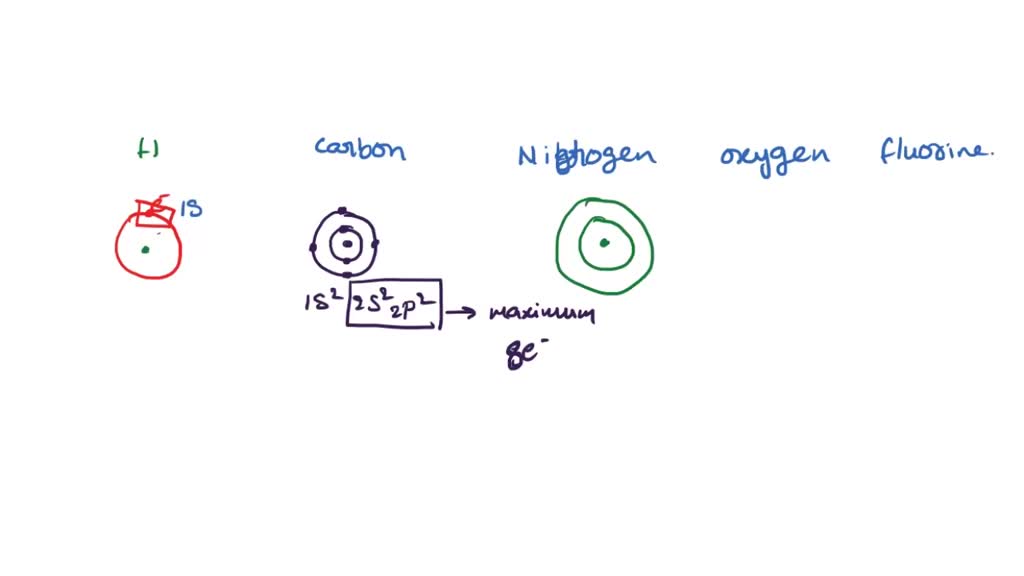
SOLVED: Given the shell model of the atom, suggest a possible reason that Lewis proposed a maximum of two electrons for hydrogen and a maximum of eight for carbon, nitrogen, oxygen, and
![PDF] Ab initio coupled-cluster effective interactions for the shell model: application to neutron-rich oxygen and carbon isotopes. | Semantic Scholar PDF] Ab initio coupled-cluster effective interactions for the shell model: application to neutron-rich oxygen and carbon isotopes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/31bd559d7f9e08daaa549531c07f38587341584e/3-Figure2-1.png)
PDF] Ab initio coupled-cluster effective interactions for the shell model: application to neutron-rich oxygen and carbon isotopes. | Semantic Scholar