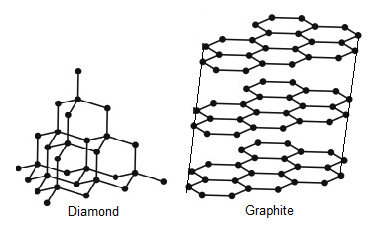
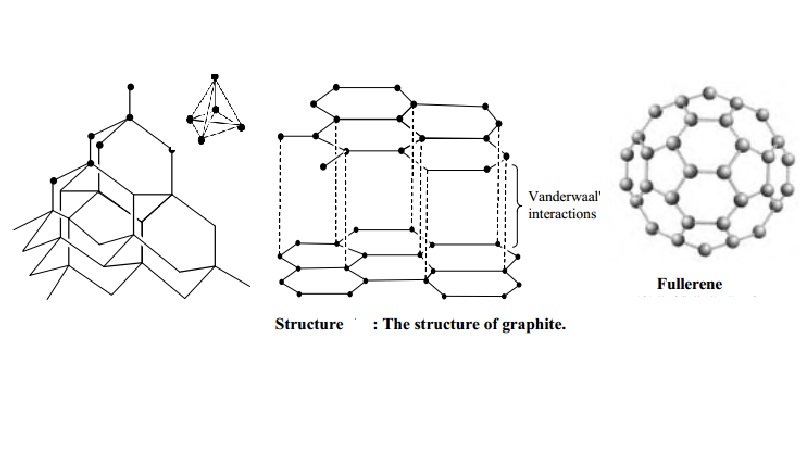
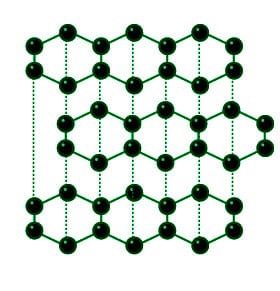
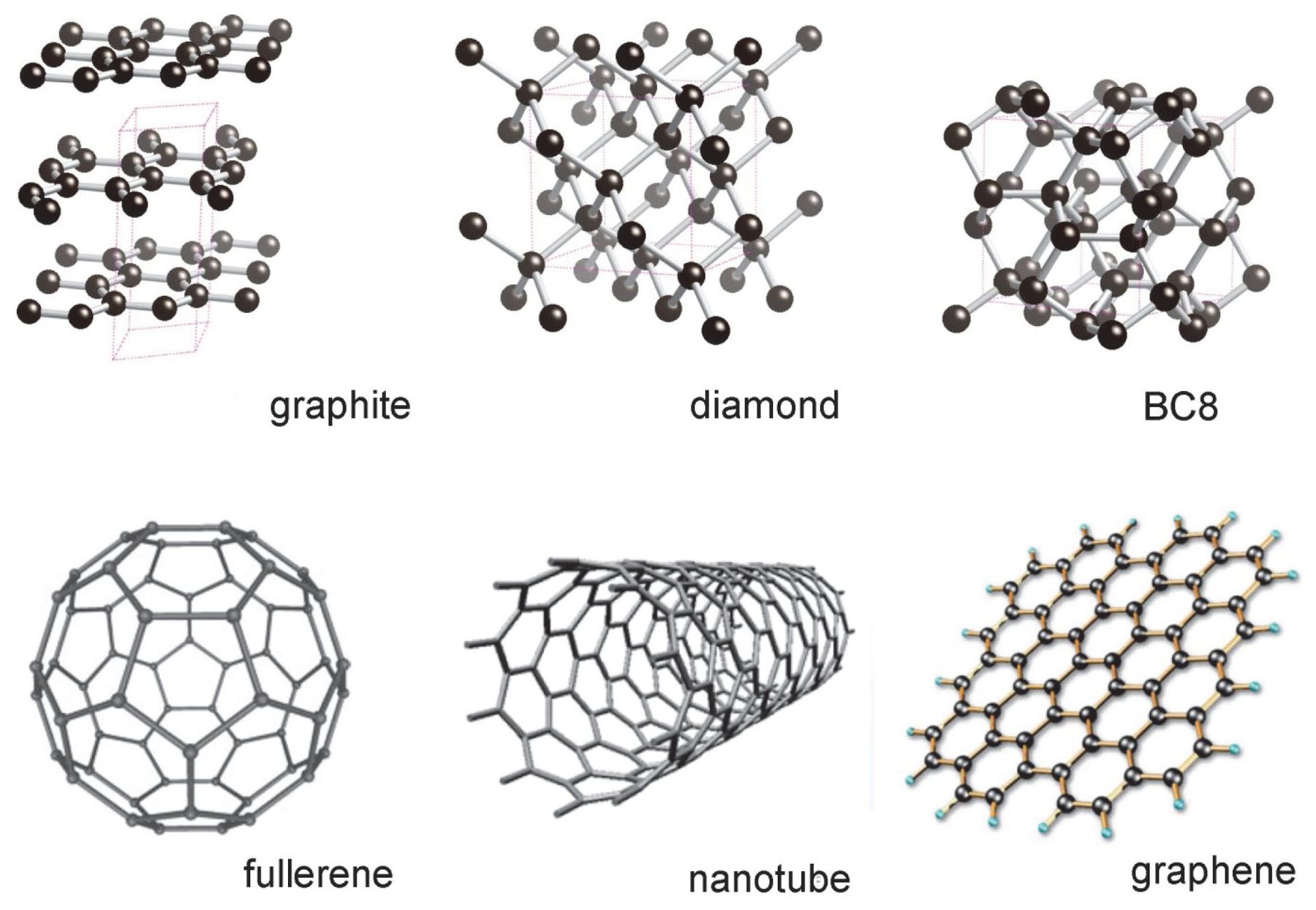
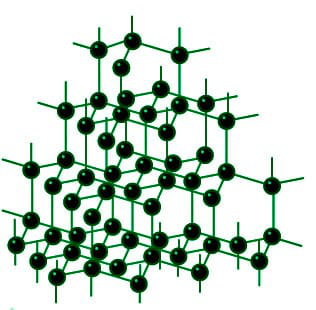
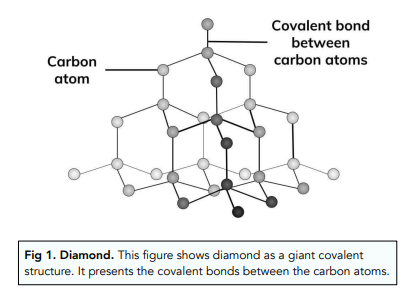
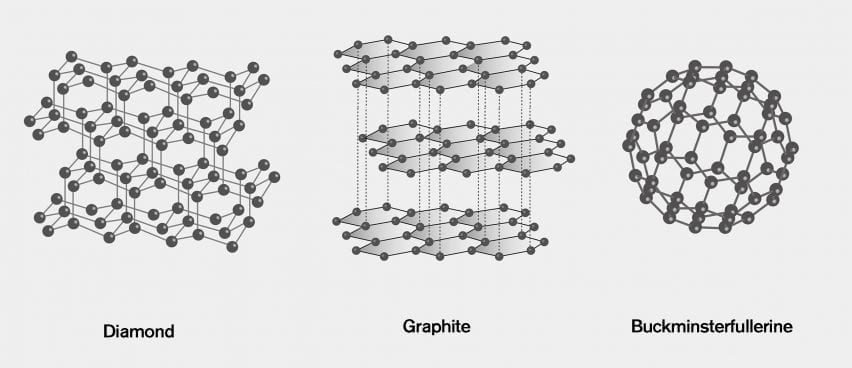
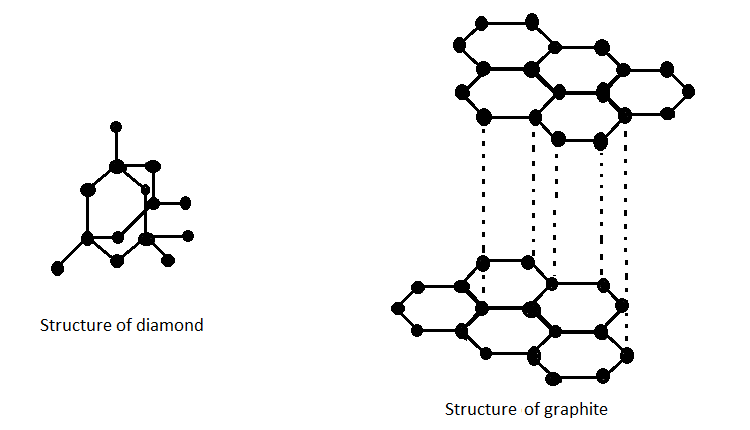
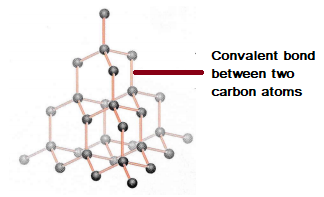
Graphite and diamond have the same composition, but how do their properties have great difference with each other like their structure, price, color, etc.? - Quora
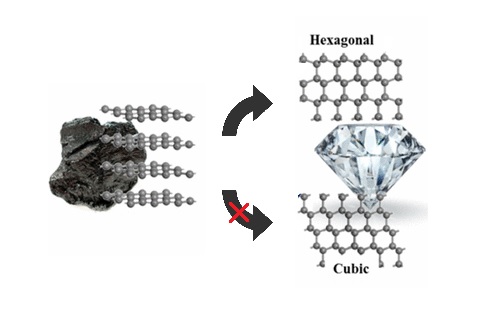
butterfly110 on Twitter: "How can graphite and diamond be so different if they are both composed of pure carbon? #Bio110Fall18 @NutmegSomething https://t.co/d70J08szHV" / Twitter

Why diamond is so shiny whereas graphite is not shiny despite the fact that both are made up of carbon? - Quora
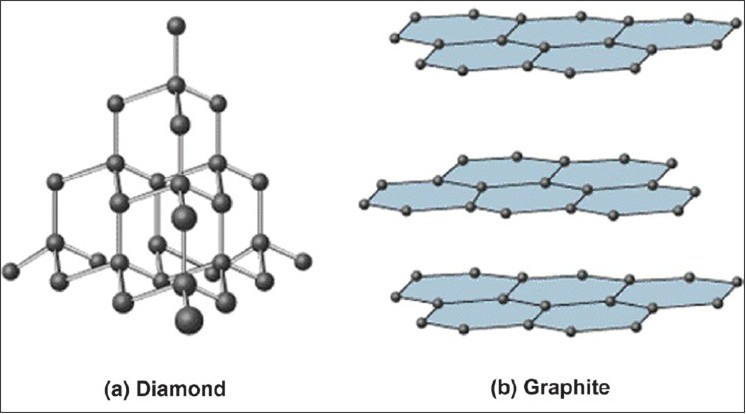
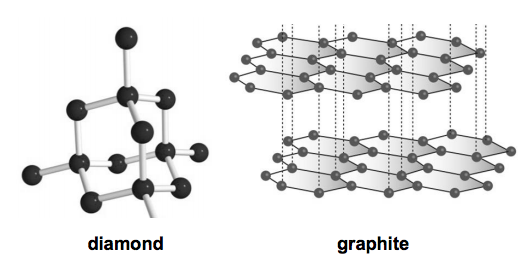
Why does the electrical conductivity of diamond and graphite differ so widely? Explain on the basis of their structure. from Science Carbon and its Compounds Class 10 Uttarakhand Board

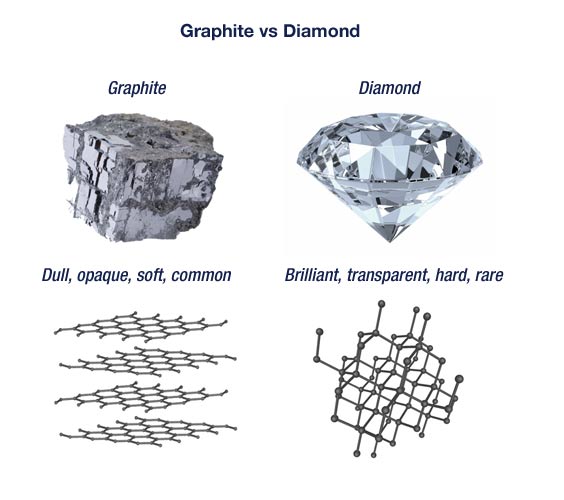
UNM Foundation Engineering - Do you know that diamond and graphite are made from the same element? Source: https://www.bbc.co.uk/bitesize/guides/zspxdxs/revision/2 . . . . . . #WeAreUoN #UoNMalaysia #UNM #nottinghammalaysia #nottinghamuniversity ...