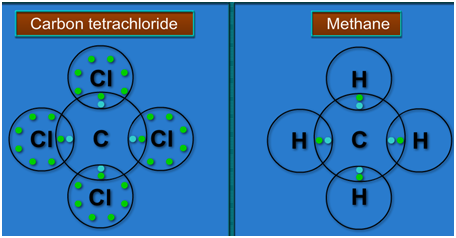
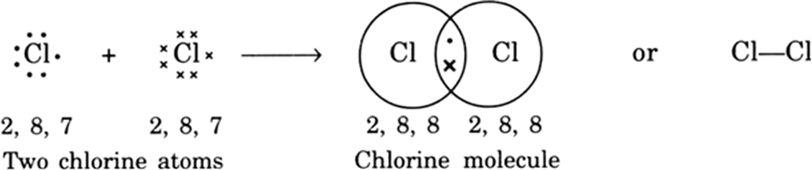Which compound among CCL4 (carbon with chlorine) and CI4 (carbon with iodine) is more covalent and why? - Quora

Explain the tetravalency of carbon with the help of electron dot structure - Science - Carbon and its Compounds - 10826407 | Meritnation.com

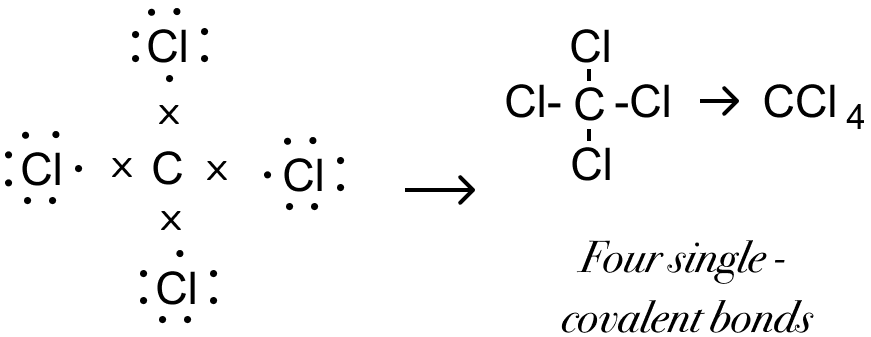
In the formation of Carbon tetra chloride molecule, state the following.a) Electronic configuration of nearest inert gas attained.b) How many electrons are sahred/ transferred in bond formation?c) Which type of bonds these

Explain the formation of Covalent compound.i) Chlorine Moleculesii) Carbon Tetra Chlorideiii) Ammonia - Brainly.in

b In the formation of carbon tetrachloride molecule, state the following:i Electronic configuration of nearest inert gas attained.ii How many electrons are shared/transferred in bond formationiii Which type of bonds these compounds

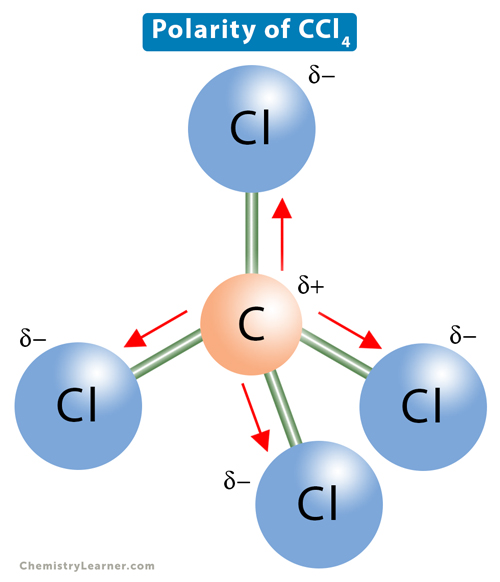
Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar