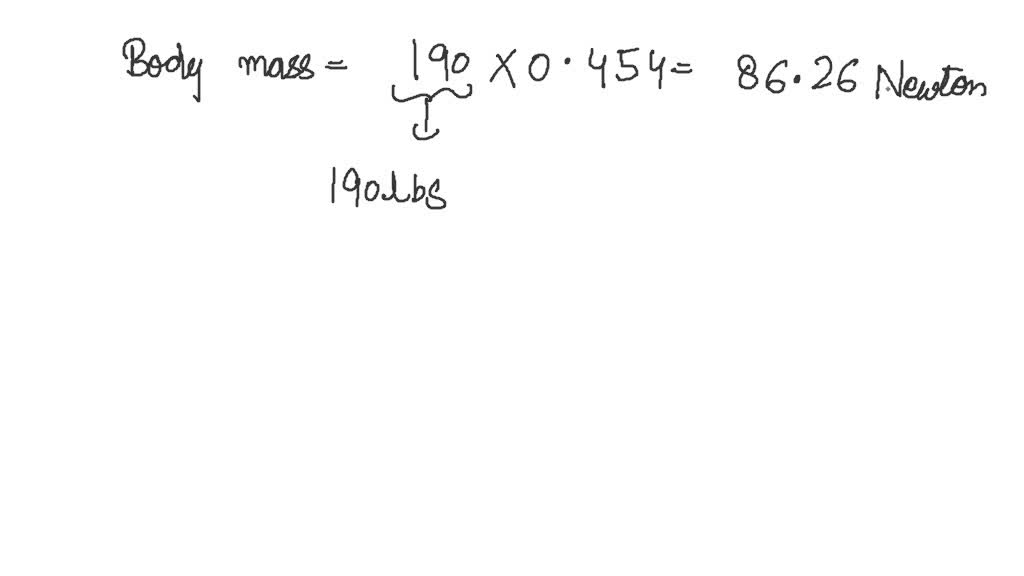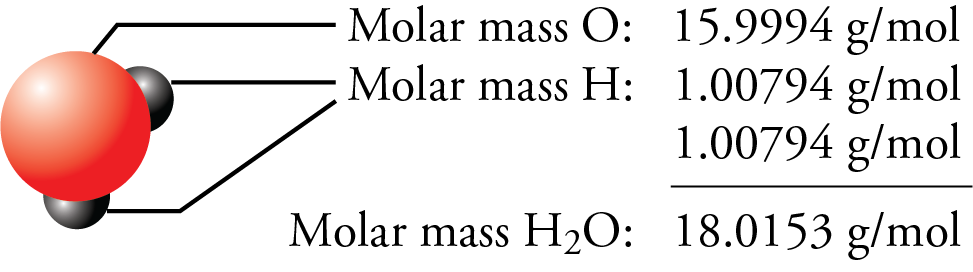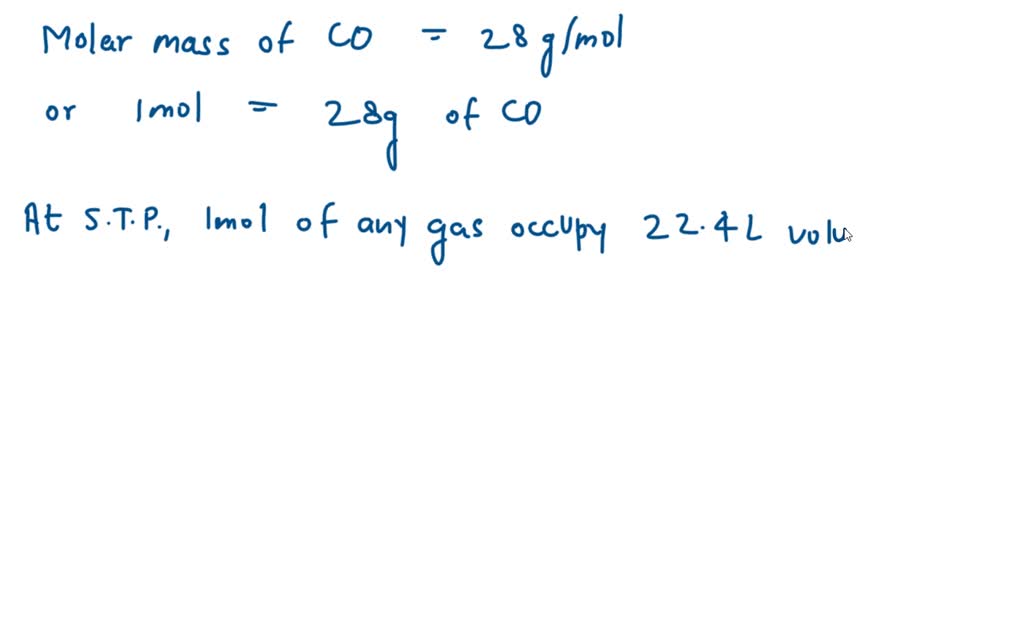
SOLVED: What is the mass in grams of 1 liter of carbon monoxide, CO, at standard temperature and pressure, STP? Note the molecular weight of CO is 28 g/mole and at STP,

Take your periodic table out. What is atomic mass of Carbon Point where you can find it in the periodic table! 6 is atomic number not atomic mass Atomic. - ppt download