Visualization of the shortest routes for the decomposition of isolated... | Download Scientific Diagram
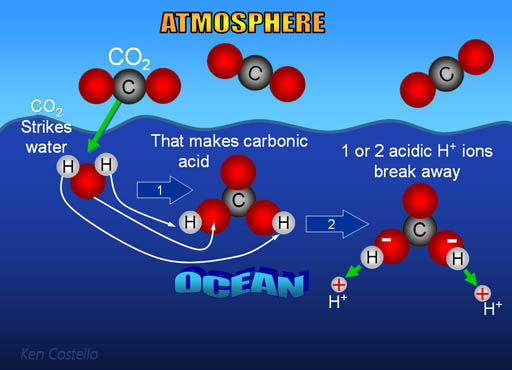
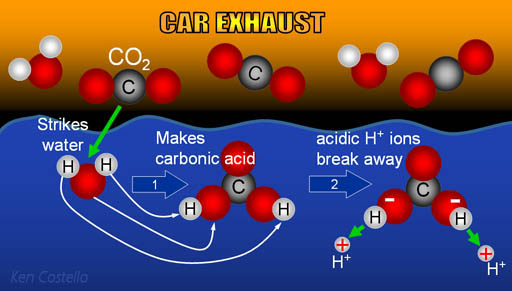
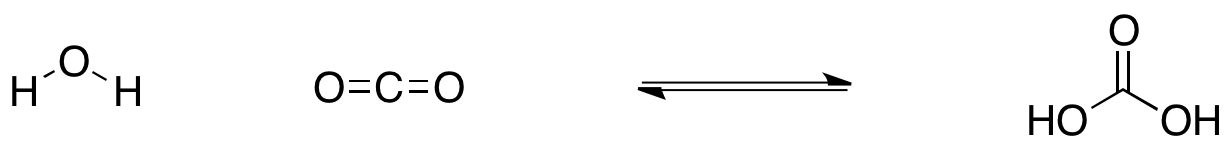
What is the most likely mechanism for the formation of carbonic acid from water and carbon dioxide? - Quora
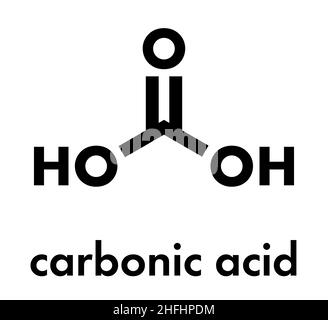
Carbonic acid molecule. Formed when carbon dioxide is dissolved in water (carbonated water). Skeletal formula Stock Vector Image & Art - Alamy
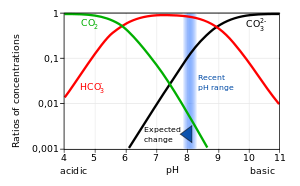
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora
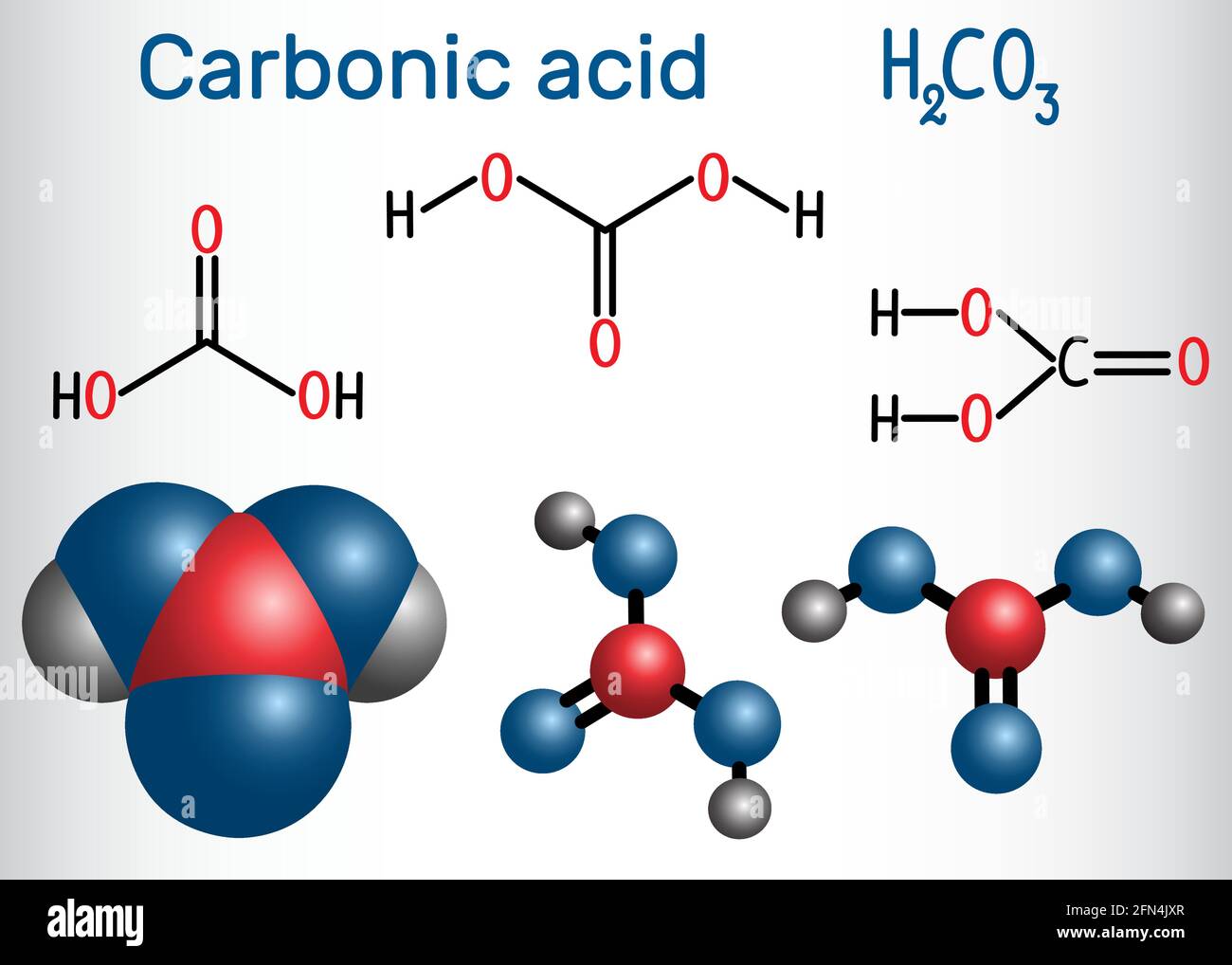
Carbonic acid (H2CO3) molecule . It is also solution of carbon dioxide in water (carbonated water). Structural chemical formula and molecule model. V Stock Vector Image & Art - Alamy

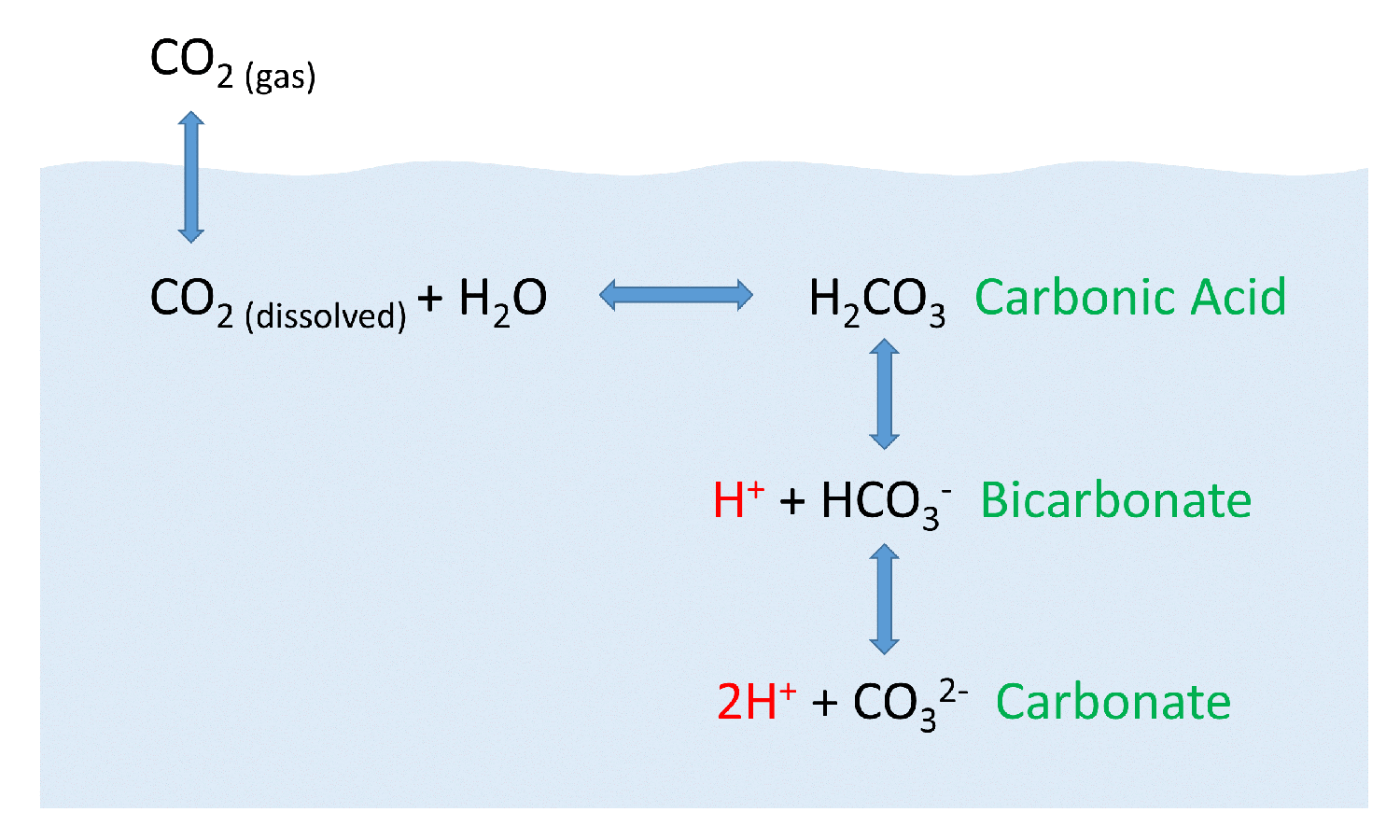
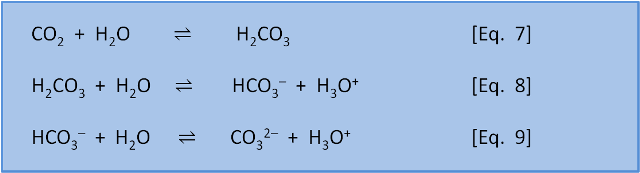
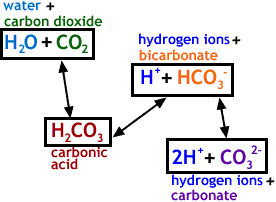
SOLVED: Carbon dioxide reacts with water in our blood to form carbonic acid. This reaction is critical in maintaining constant pH values Water Hydrogen ion HzCOz H+ HCO3 Carbonic Bicarbonate acid ion
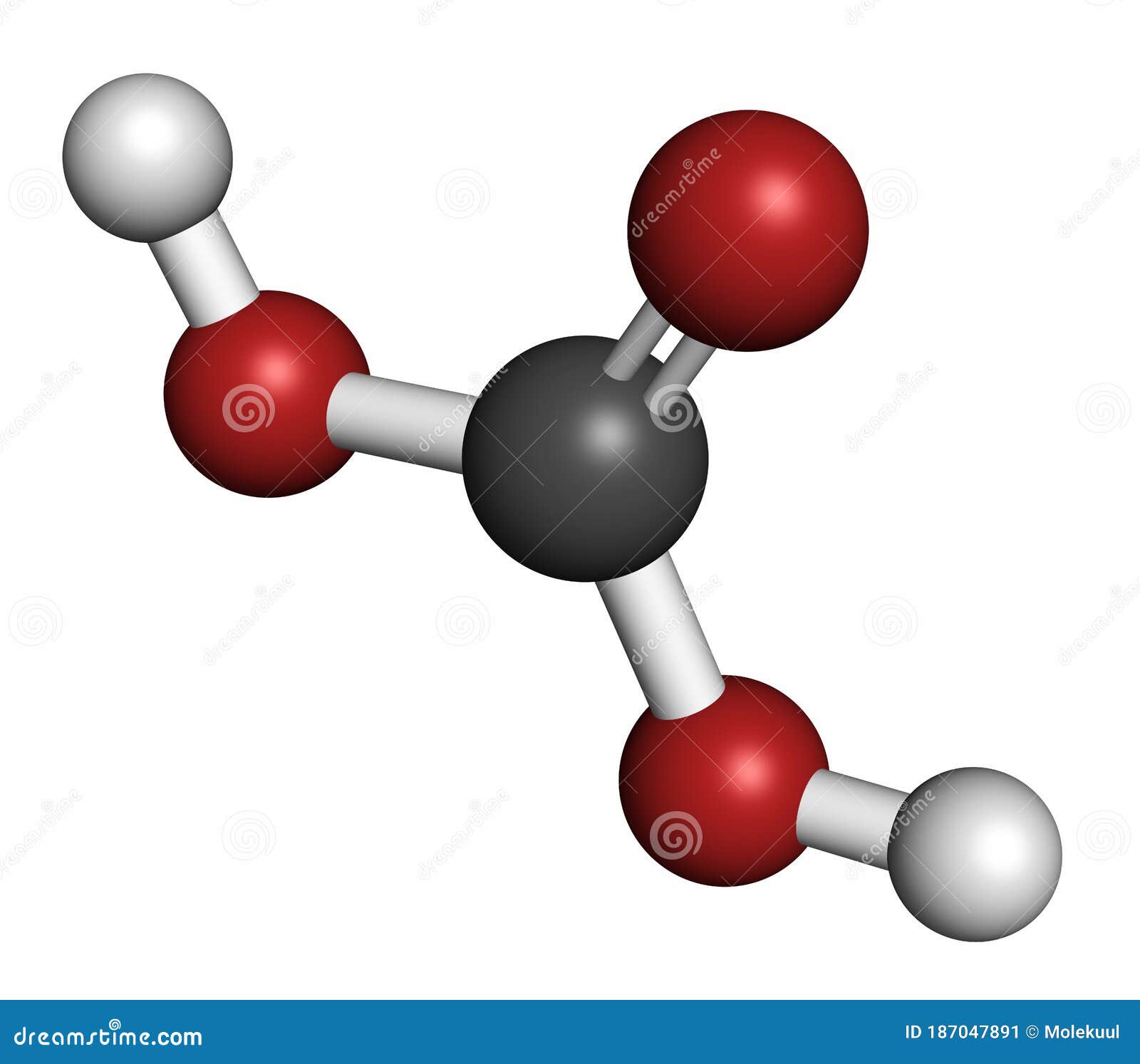
Carbonic Acid Molecule. Formed when Carbon Dioxide is Dissolved in Water (carbonated Water). 3D Rendering Stock Illustration - Illustration of formula, atomic: 187047891