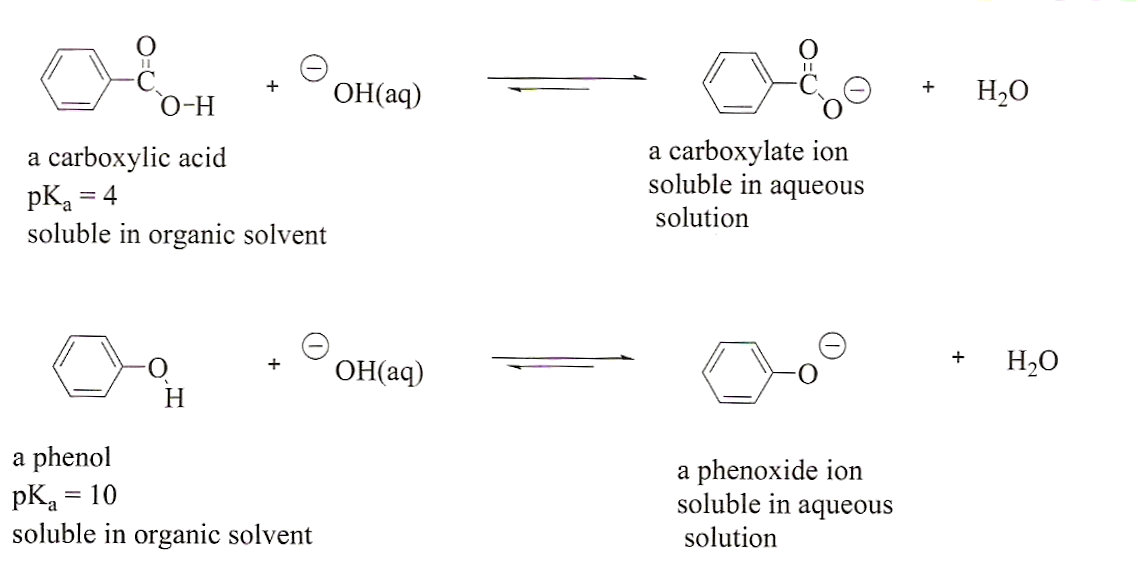
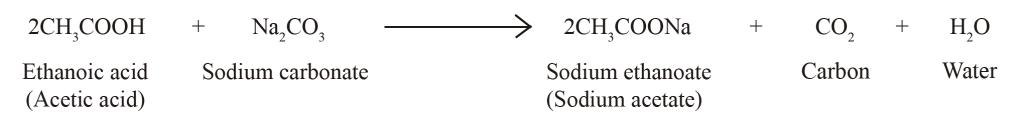The reaction of sodium hydrogen carbonate with ethanoic acid is used as a test for detection of carboxylic acid, why?

SOLVED: Na,CO, Carboxylic acids react with sodium carbonate t0 form the sodium salt of the carboxylic acid, watcr and carbon dioxide Fizzing in the reaction can be viewed as # positive test




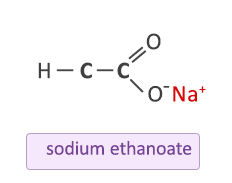
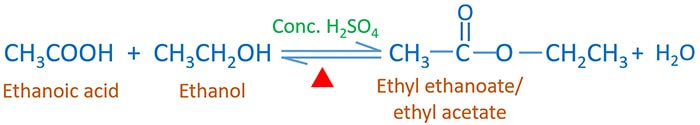

![Carbon] Ethanoic acid - Formation, Properties, Uses [with Reactions] Carbon] Ethanoic acid - Formation, Properties, Uses [with Reactions]](https://d1avenlh0i1xmr.cloudfront.net/fb1b0fbe-d74a-44bb-8d4d-1b41b5aa53a2/reaction-of-ethanoic-acid-with-sodium-carbonate-and-sodium-bicarbonate---teachoo.jpg)