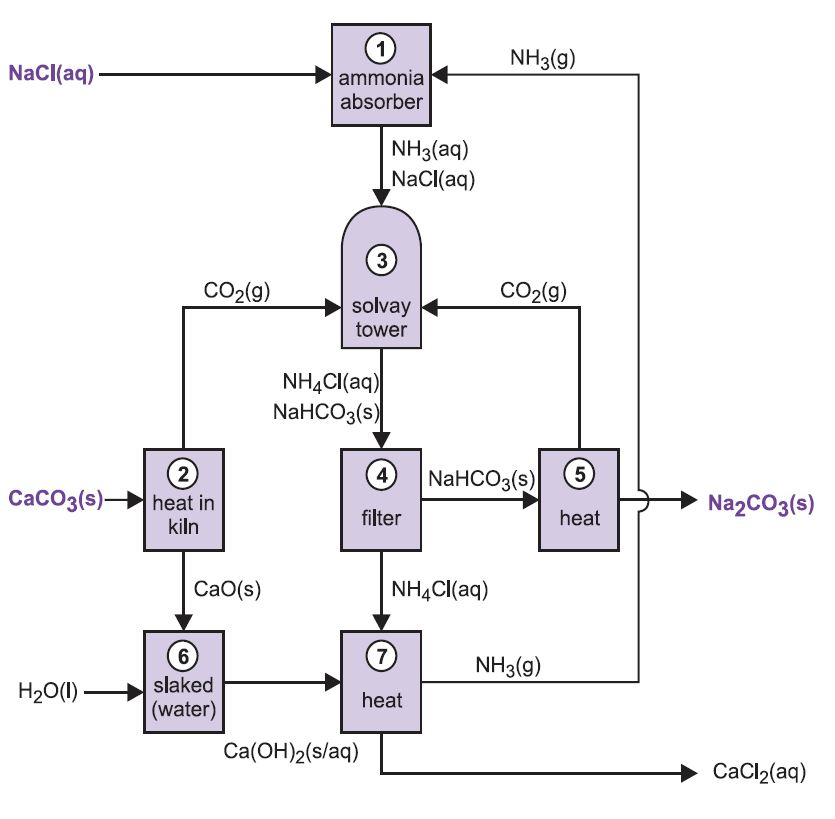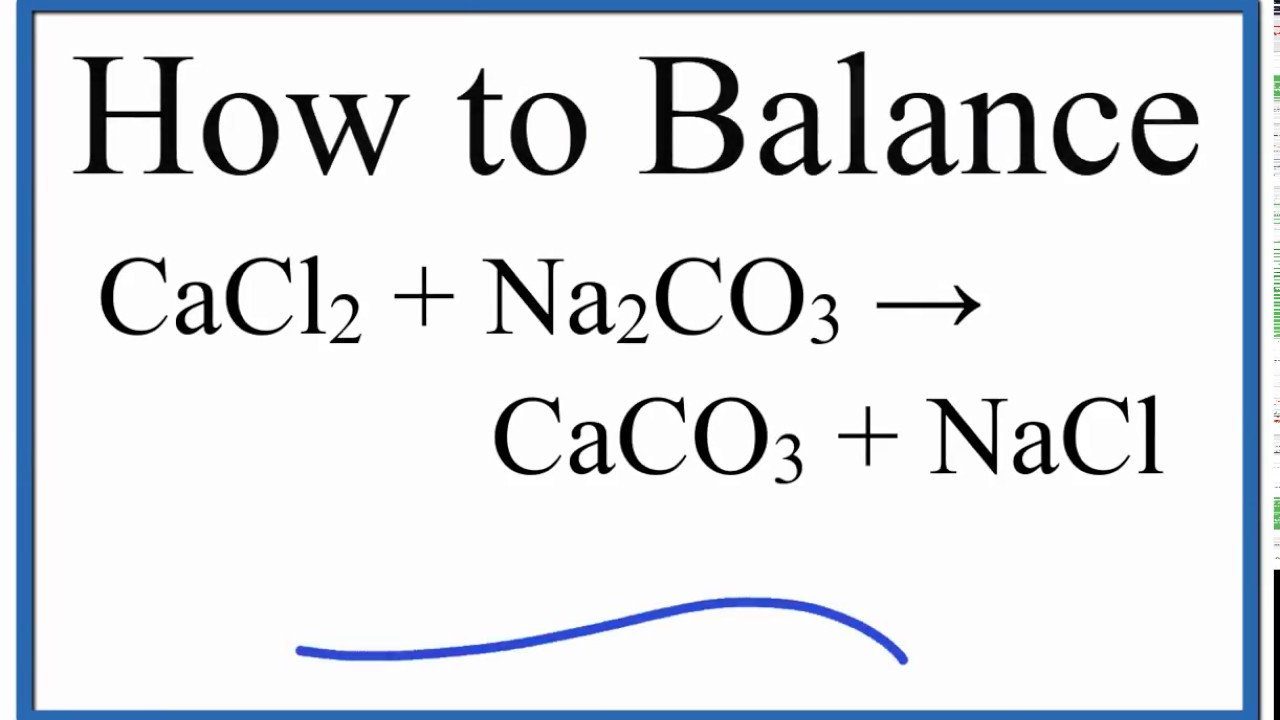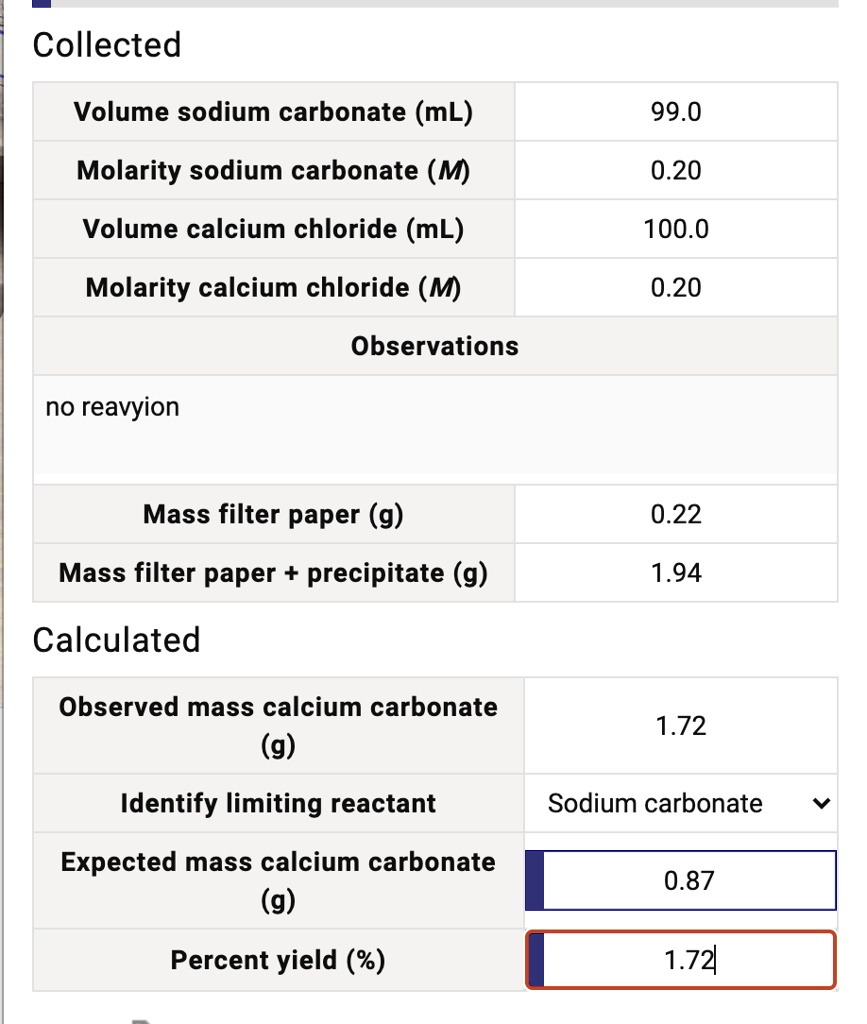
SOLVED: Collected Volume sodium carbonate (mL) 99.0 Molarity sodium carbonate (M) 0.20 Volume calcium chloride (mL) Molarity calcium chloride (M) 100.0 0.20 Observations no reavyion Mass filter paper (g) 0.22 Mass filter
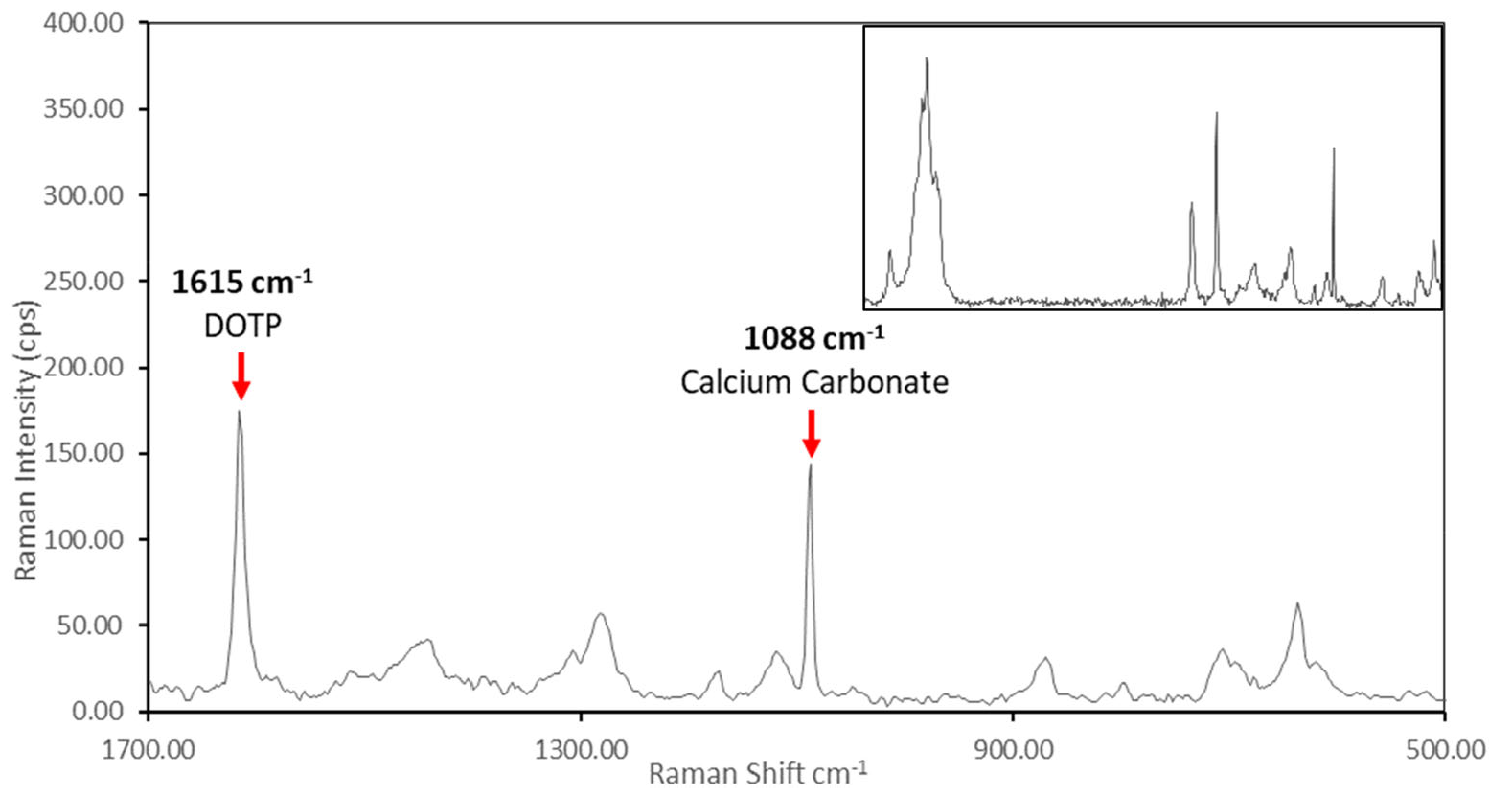
The quantification of di-octyl terephthalate and calcium carbonate in polyvinyl chloride using Fourier transform-infrared and Raman spectroscopy – Defrosting the Freezer
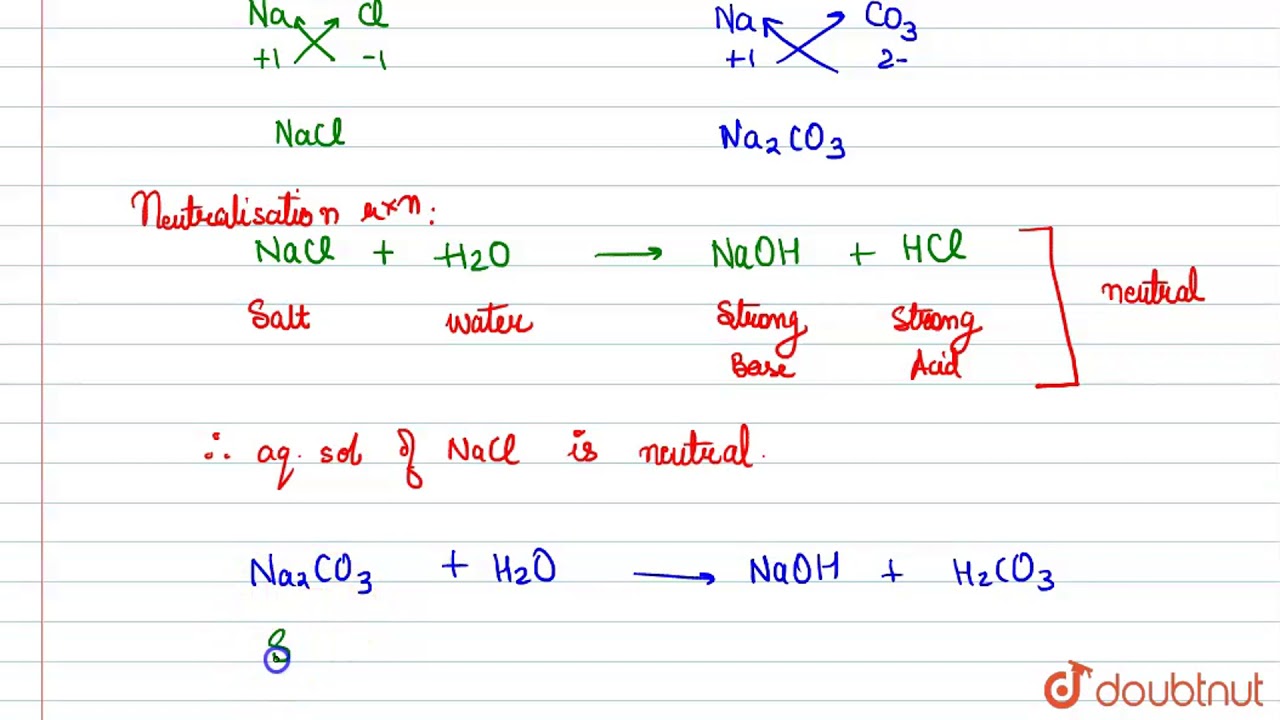
Write the formulae of sodium chloride and sodium carbonate. Explain why an aqueous solution of - YouTube
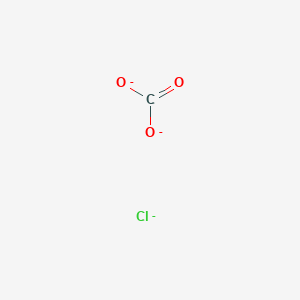
111 gram sample of a mixture of calcium chloride and Sodium Chloride is treated with sodium carbonate to precipitate precipitate as Calcium precipitate this kkcalcium carbonate on heating gives 14 gram of

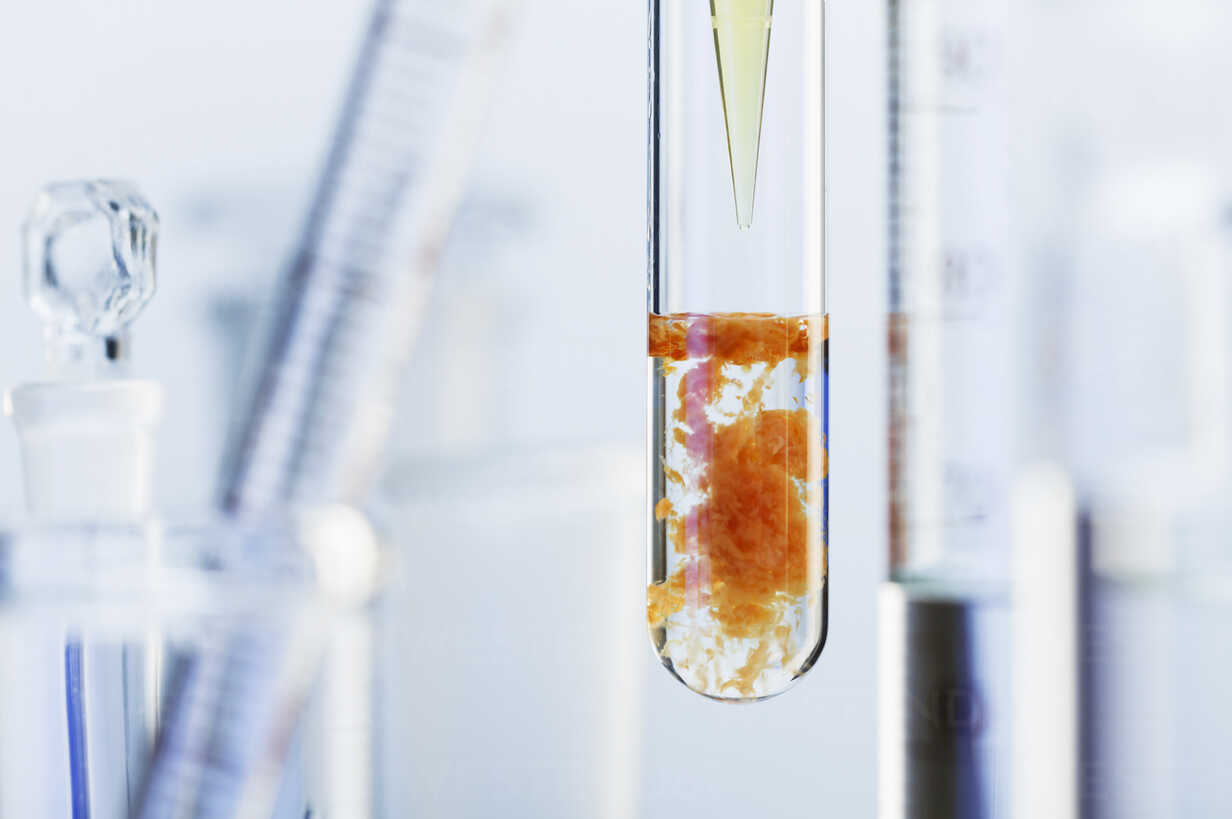
What happens when aqueous solutions of calcium chloride and of sodium carbonate are mixed? | Socratic
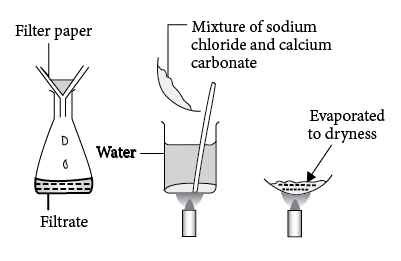
A mixture of sodium chloride and calcium carbonate is separated using the steps given. In which order should the steps be carried out?\n \n \n \n \n A.1,2 then 3B.2,1 then 3C.1,3

Composition inversion to form calcium carbonate mixtures - CrystEngComm (RSC Publishing) DOI:10.1039/C7CE00433H