Collection of molecular chemical models combinations from hydrogen oxygen sodium carbon nitrogen and chlorine. Vector molecules set illustration isolated on white background Stock Vector Image & Art - Alamy

Chloroform molecule. The center black sphere is a carbon atom which is... | Download Scientific Diagram
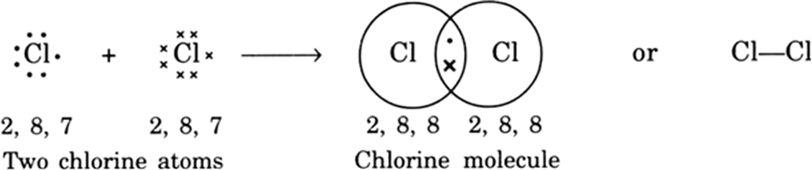
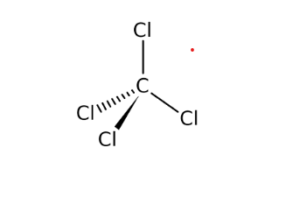
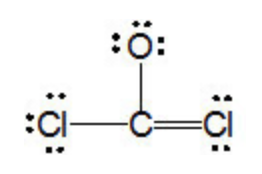
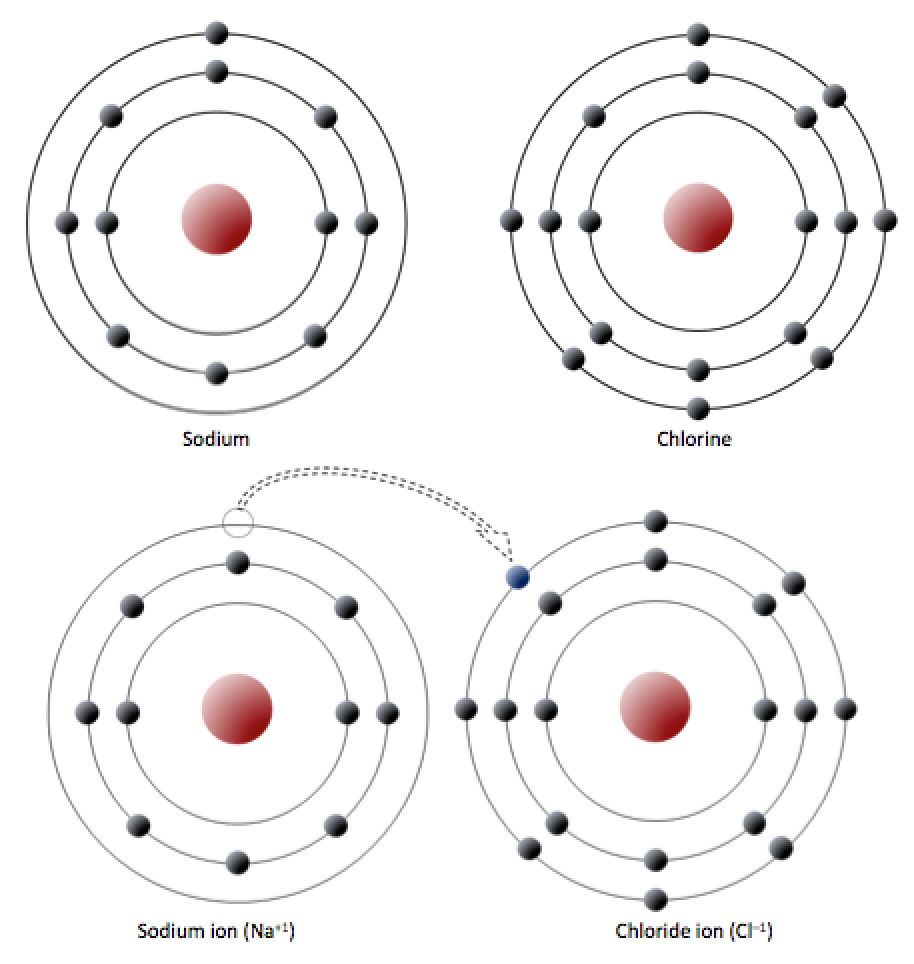
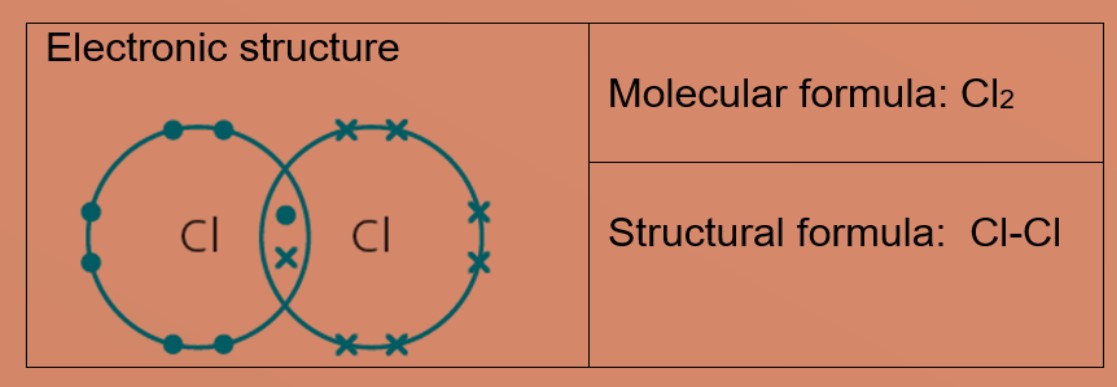
Explain the formation of covalent bonds in (i) chlorine molecule; (ii) carbon tetrachloride and (iii) ammonia. from Science Carbon and its Compounds Class 10 Jammu and Kashmir Board

Diagrammatic summary of the procedure for carbon and chlorine stable... | Download Scientific Diagram

Carbon, Hydrogen and Chlorine Stable Isotope Fingerprinting for Forensic Investigations of Hexachlorocyclohexanes | Environmental Science & Technology
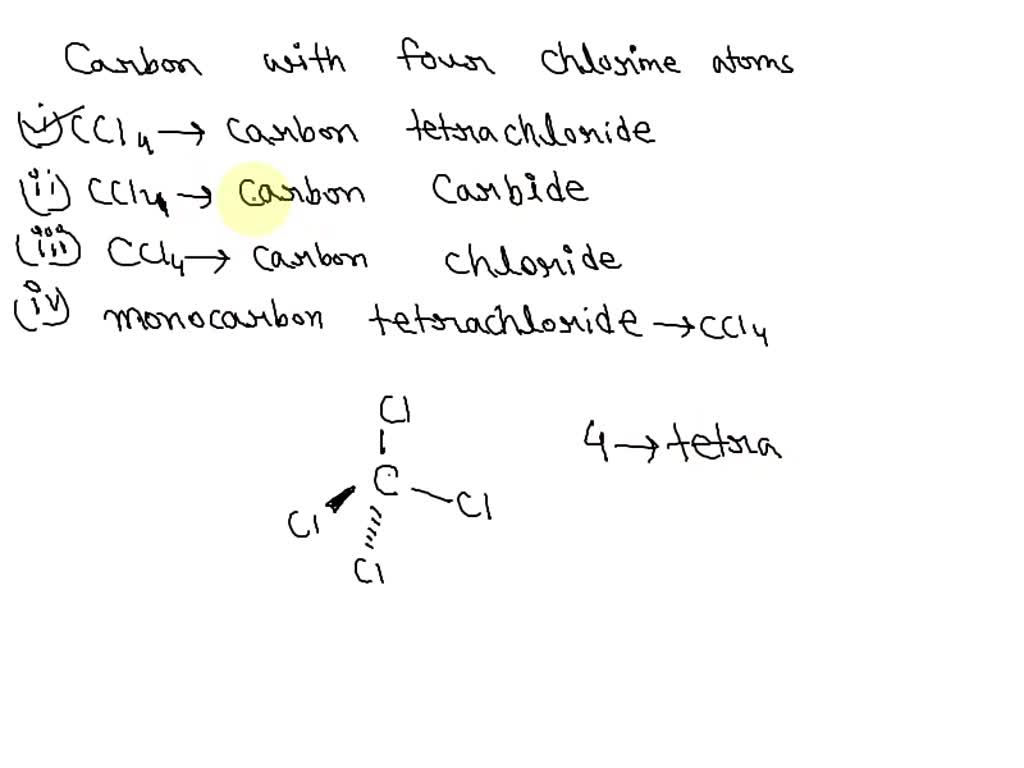
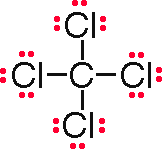
SOLVED: What is the chemical name and formula of a compound that has one carbon atom and four chlorine atoms? carbon tetrachloride; CCl4 chlorine carbide; Cl4C carbon chloride; Cl4C monocarbon tetrachloride; CCl4
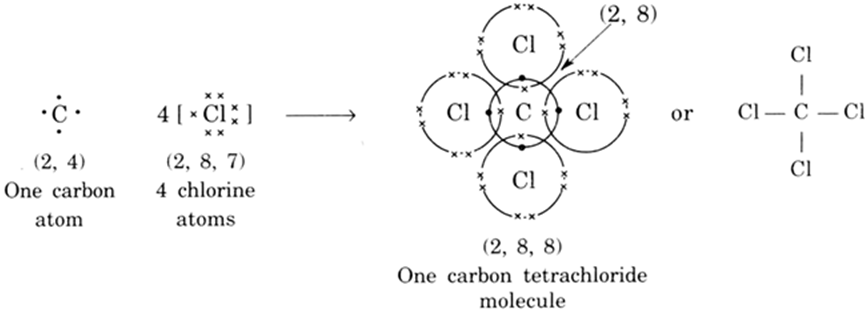
Explain the formation of covalent bonds in (i) chlorine molecule; (ii) carbon tetrachloride and (iii) ammonia. from Science Carbon and its Compounds Class 10 Jammu and Kashmir Board

Carbon reacts with chlorine to form CCl4 . 36 g of carbon was mixed with 142 g of Cl2 . Calculate the mass of CCl4 produced and the remaining mass of reactant.

Explain the formation of Covalent compound.i) Chlorine Moleculesii) Carbon Tetra Chlorideiii) Ammonia - Brainly.in

Chlorine Cl 2 Hydrogen chlorideHCl Methane CH 4 Carbon dioxide CO 2 Draw a line between the molecule and its name. - ppt download

How many chlorine atoms have to combine with a carbon atom to complete its octet during the formation of carbon tetrachloride
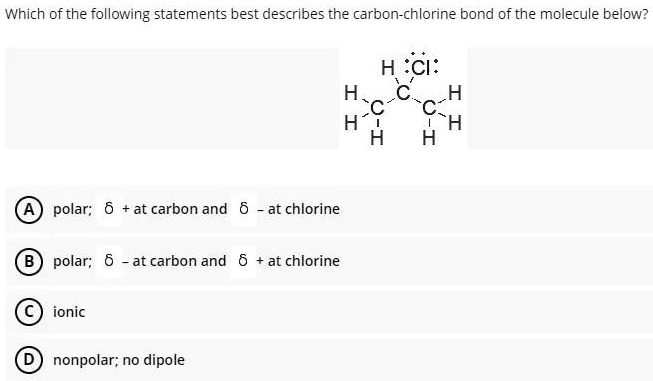
SOLVED: Which of the following statements best describes the carbon-chlorine bond of the molecule below? H :Cl: H C C- H C H H H polar; 6 +at carbon and at chlorine
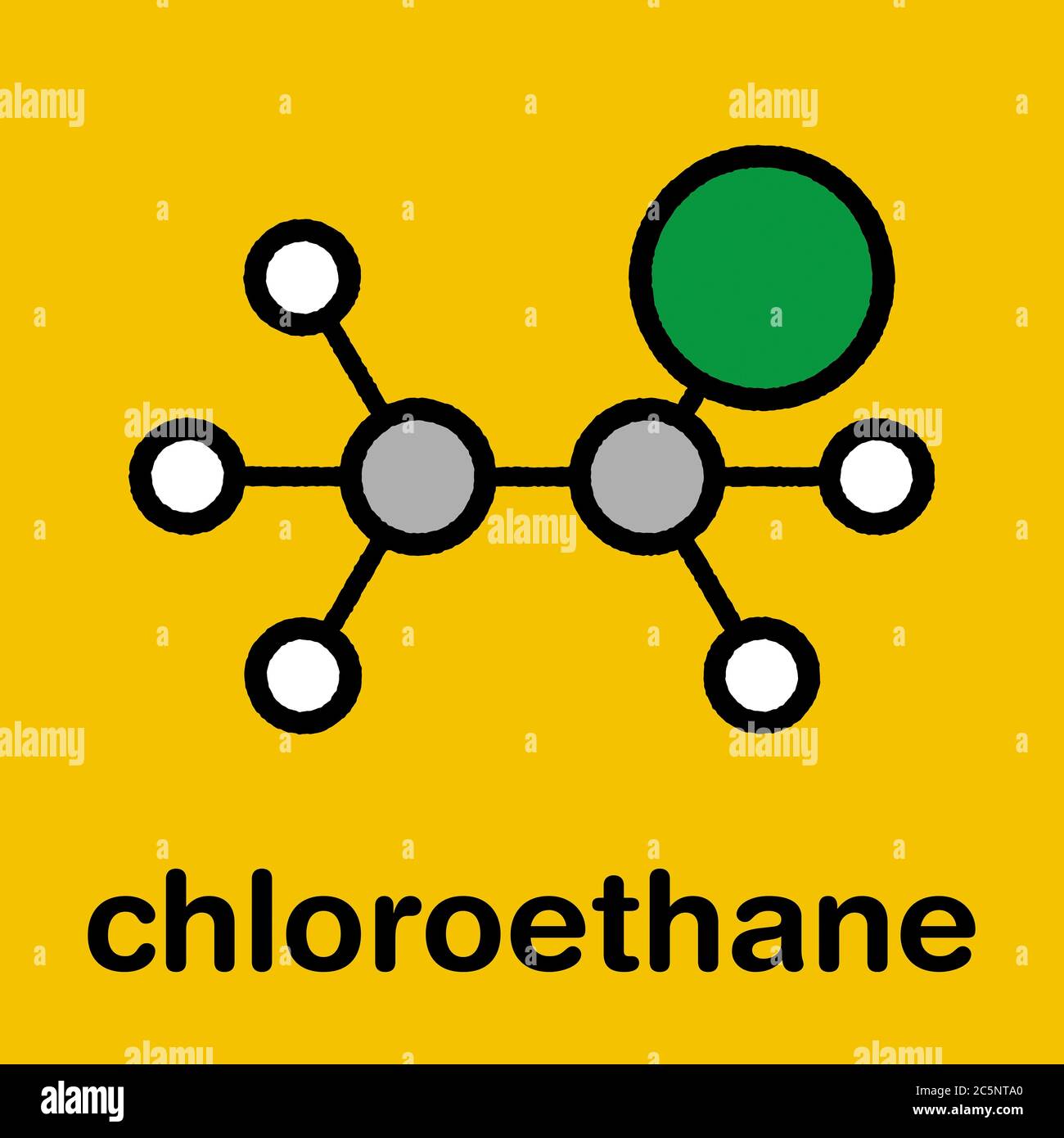
Chloroethane (ethyl chloride) local anaesthetic molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (white), carbon (grey), chlorine (green Stock Photo - Alamy

Catalytic Carbon–Chlorine Bond Activation by Selenium‐Based Chalcogen Bond Donors - Wonner - 2017 - Chemistry – A European Journal - Wiley Online Library