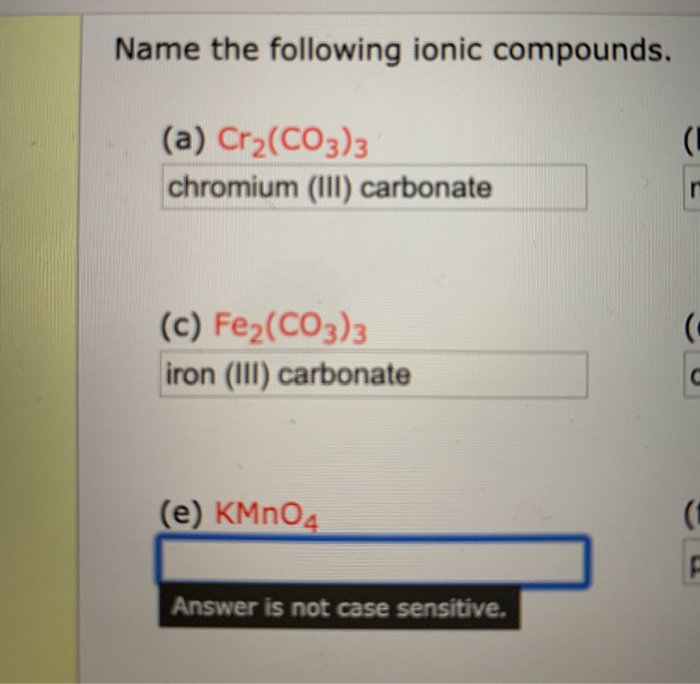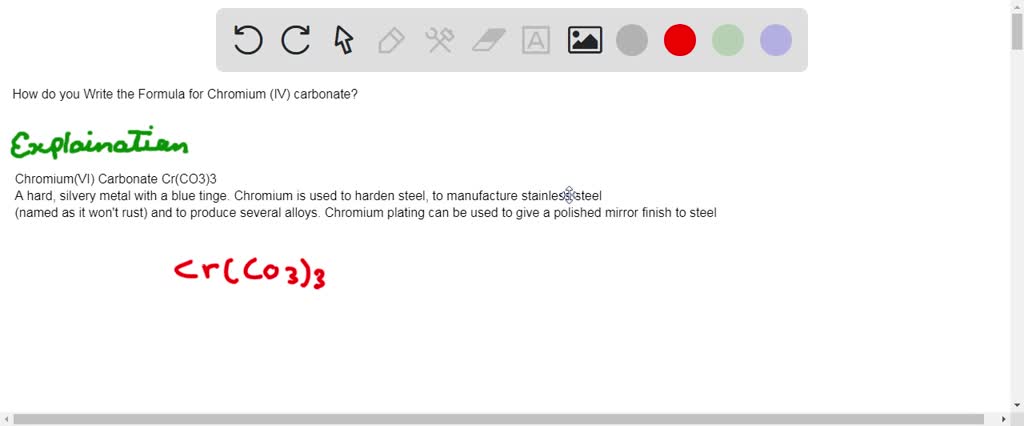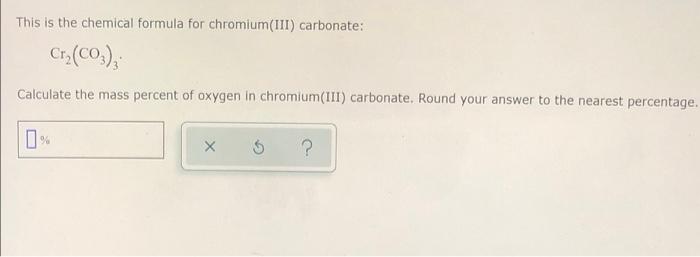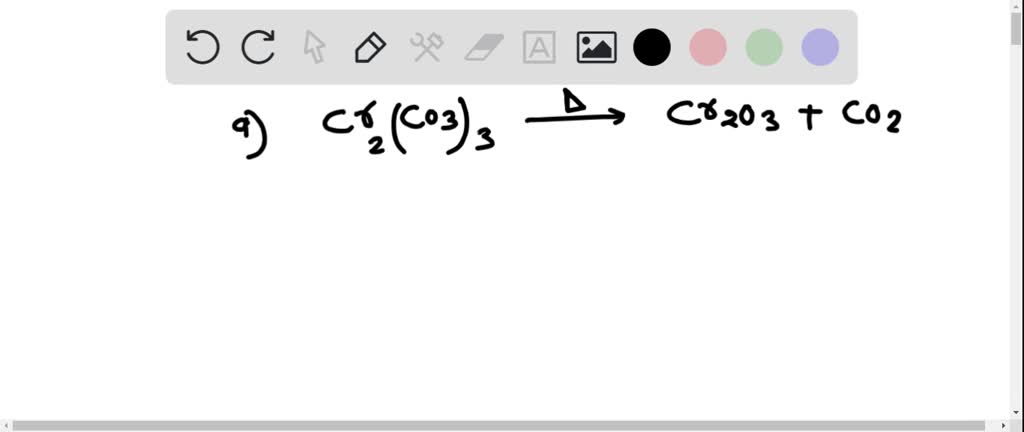
SOLVED:Write a balanced equation for each of the following decomposition reactions: (a) Chromium(III) carbonate decomposes with heat to give solid chromium(III) oxide and carbon dioxide gas. (b) Lead(IV) carbonate decomposes with heat

SOLVED: 1. Write the chemical formula for chromium(III) cyanide? 2. Write the chemical formula for chromium(III) phosphate? 3. Write the chemical formula for iron(III) carbonate 4. Write the chemical formula for nickel(II)

Warm Up Naming Ions: Aluminum IonCrO 4 2- Ammonium ionBromide ion Ba +2 Carbonate ion Cr +2 Cl - Chromium (III) ionOxide ion Cobalt (II) ionN 3- Cu + Fluoride. - ppt download
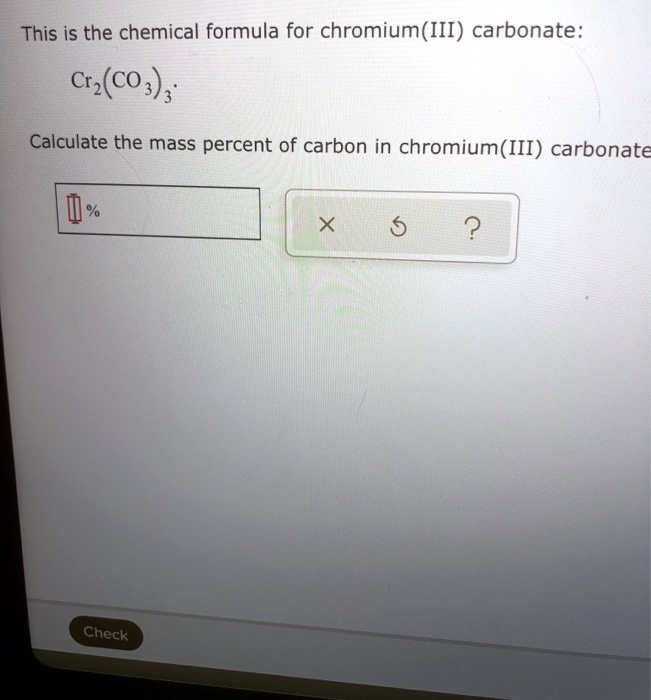
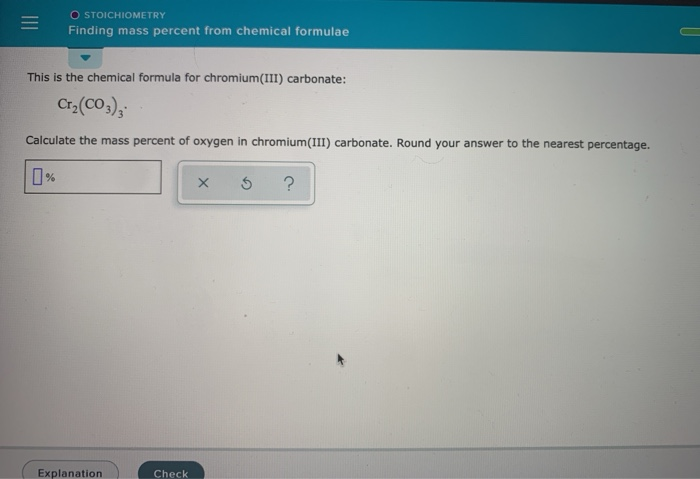
SOLVED: This is the chemical formula for chromium(III) carbonate: Cr(Cos) ; Calculate the mass percent of carbon in chromium(III) carbonate Check