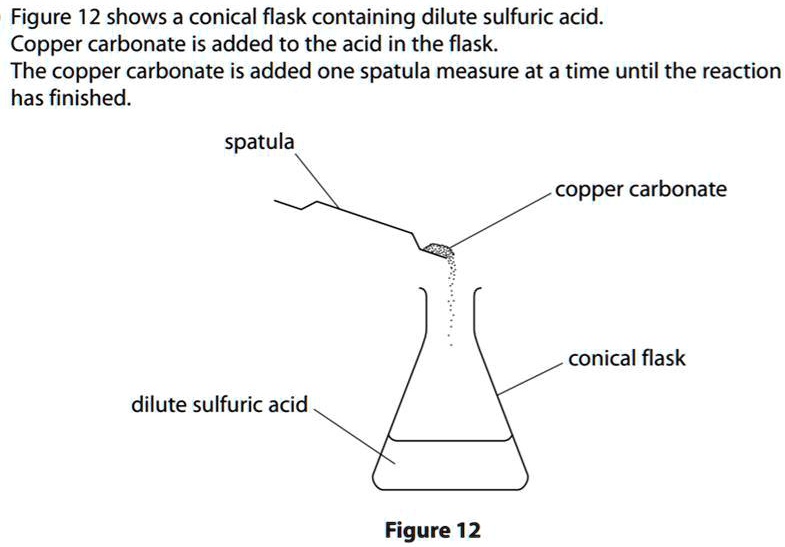
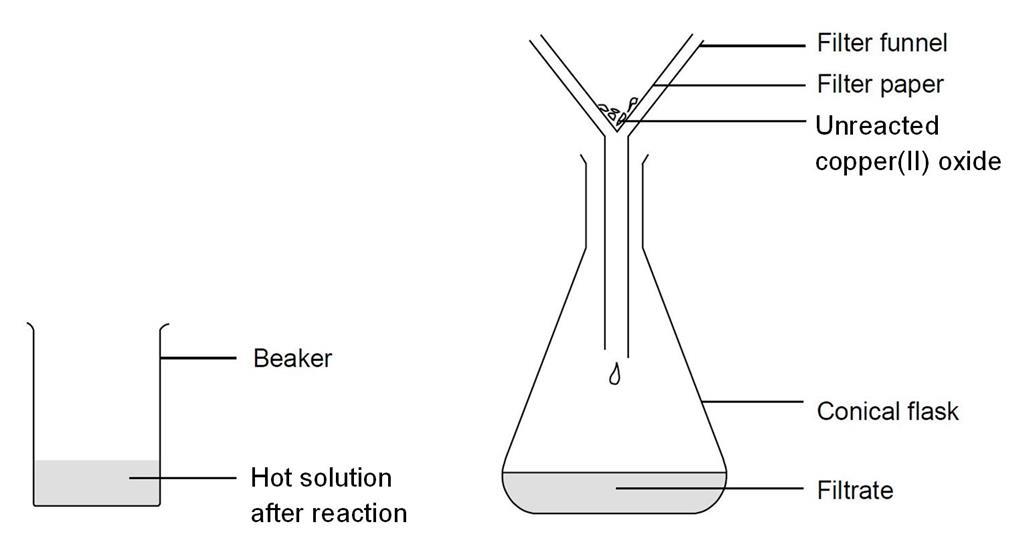
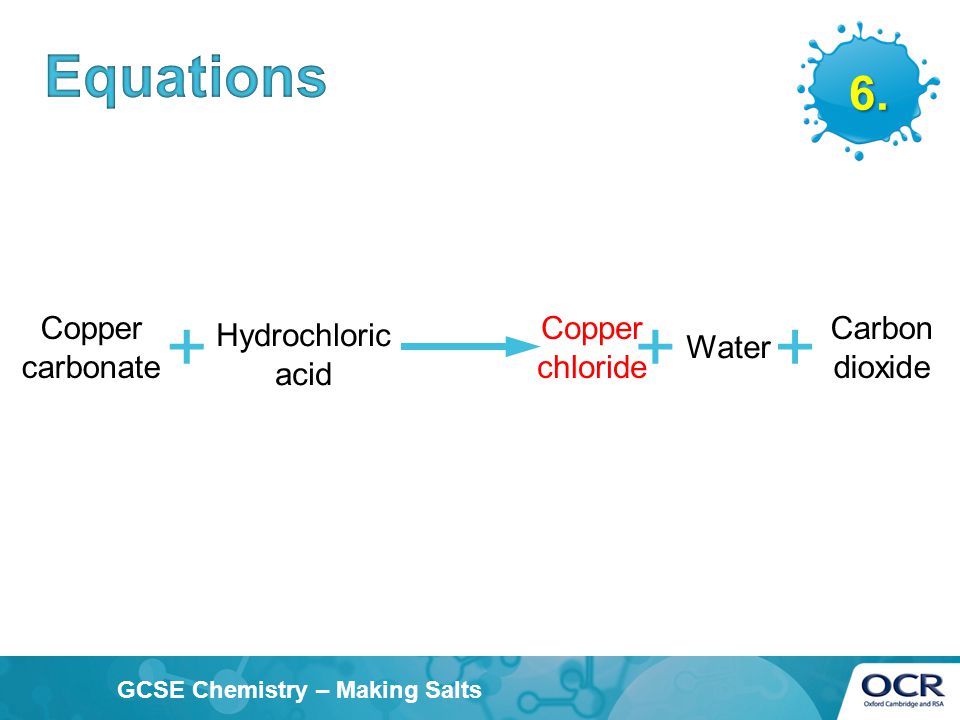
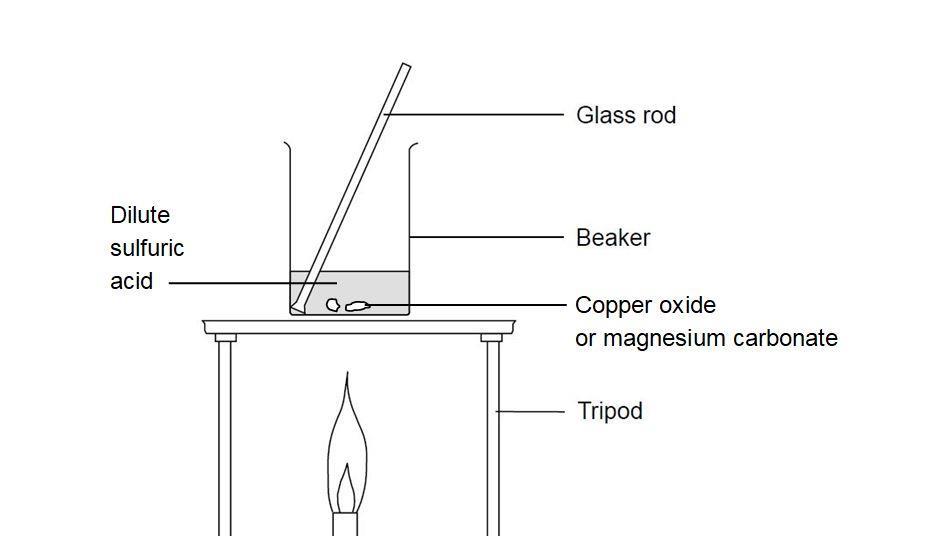
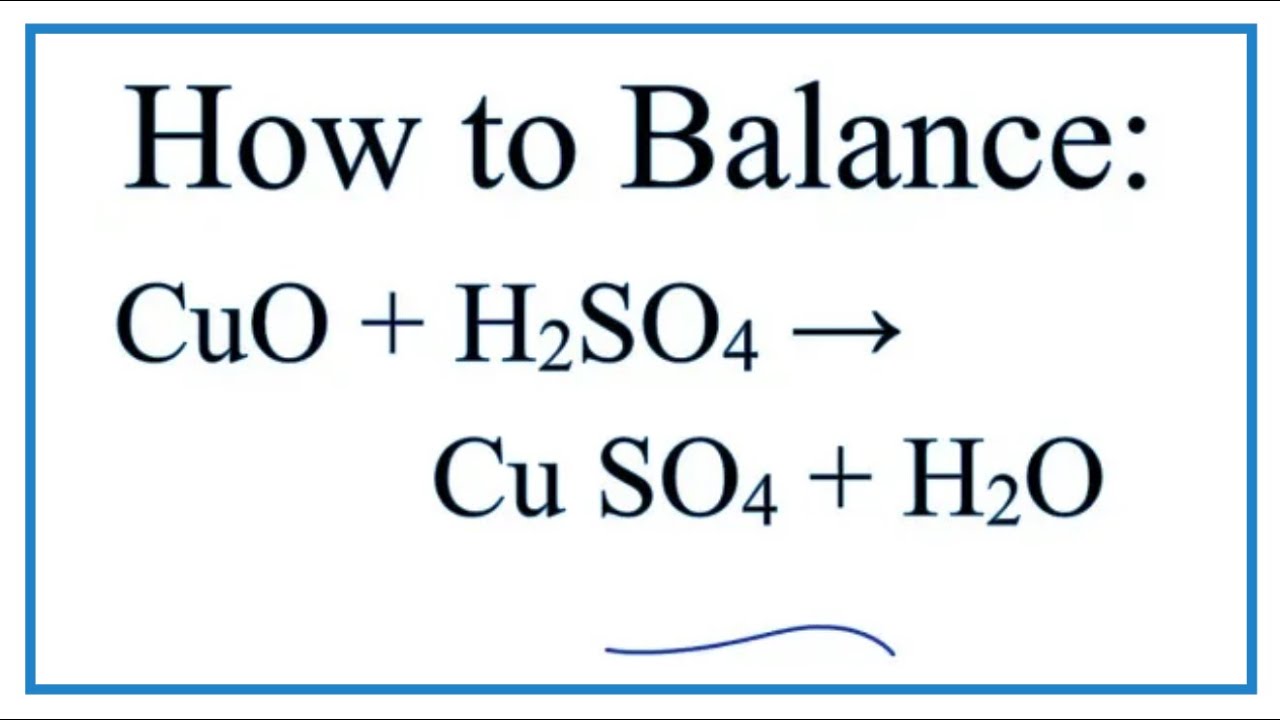
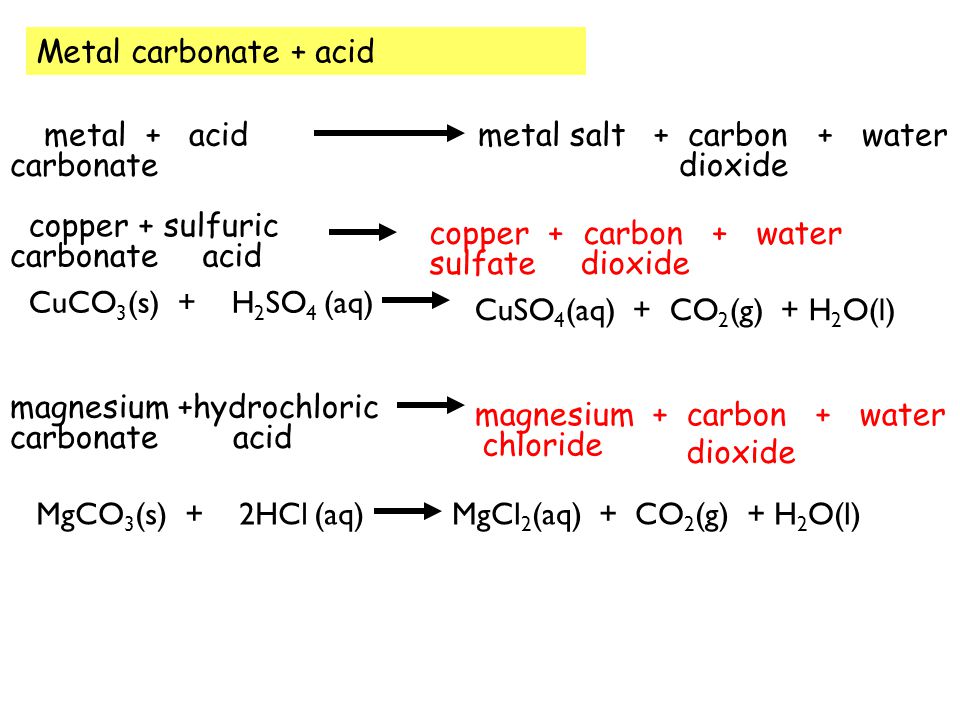
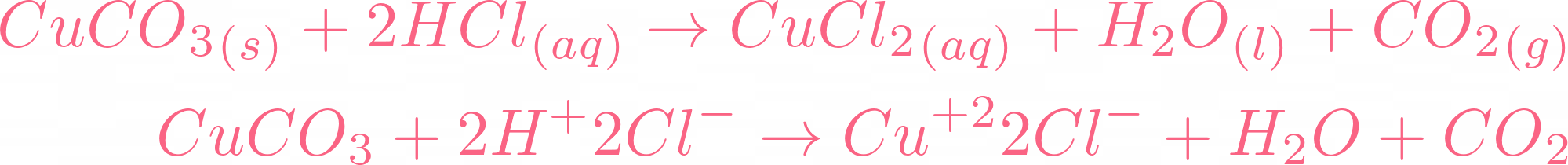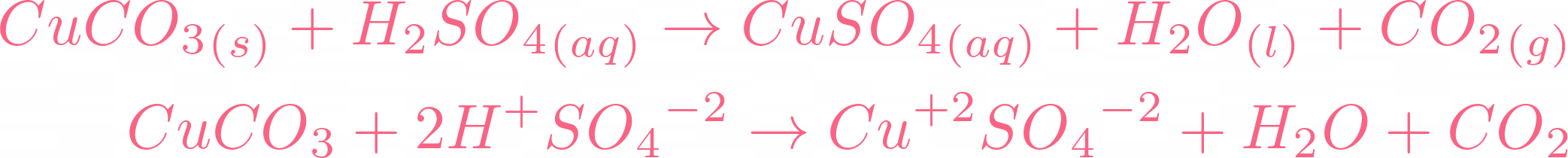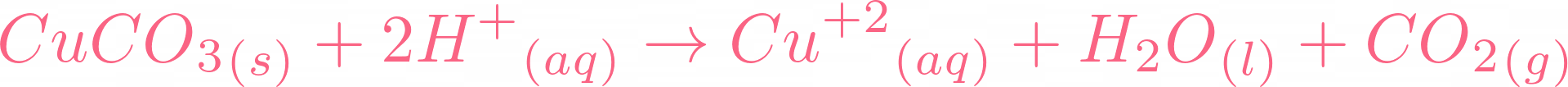i) Lead sulphate from lead carbonate (ii) Sodium sulphate using dilute sulphuric acid. (iii) Copper chloride using copper carbonate. - Sarthaks eConnect | Largest Online Education Community

How would you write the name of the following compounds ◦ Zn(OH) 2 ◦ NaOH ◦ HCl ◦ Mg(NO 3 ) 2 What does an acid do to red litmus? What does an acid to. - ppt download

Colour changes for simple indicators IndicatorColour in acidColour in alkali litmusredblue methyl orangeredyellow phenolphthaleincolourlessred. - ppt download

Choosing the substances from the list given below, write balanced chemical equations for the reaction which would be used in the laboratory to obtain the following salts. [Dilute sulphuric acid, Copper, Copper(II)
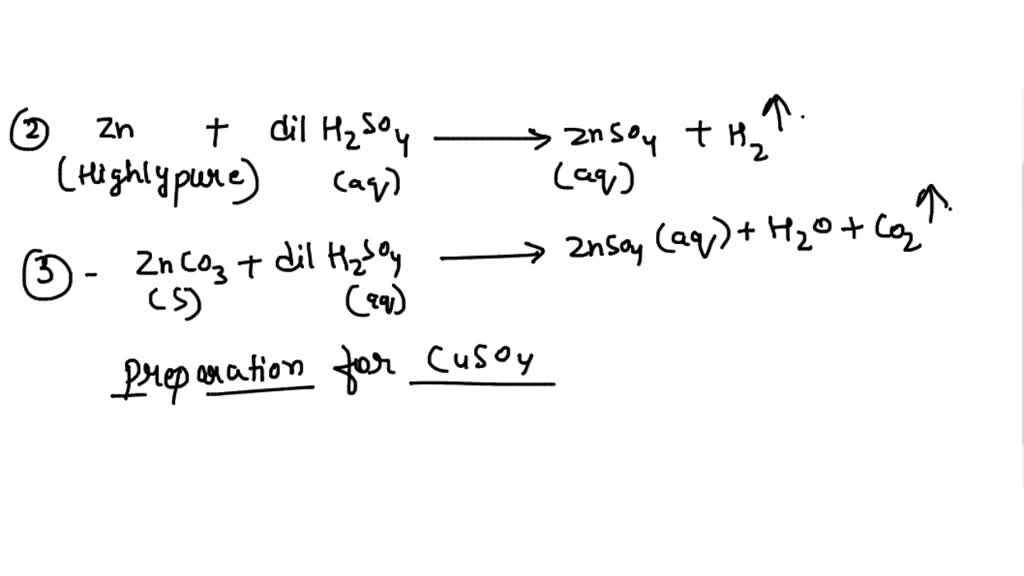
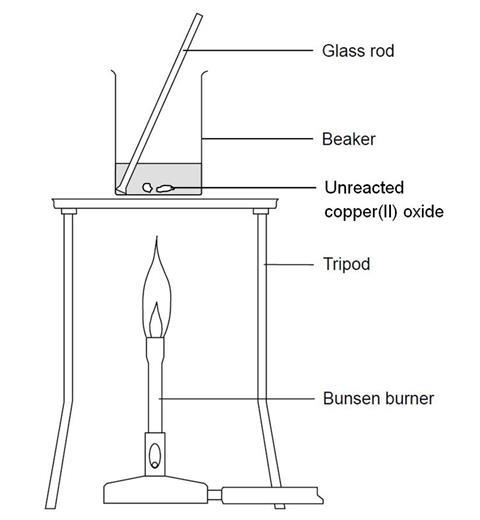
SOLVED: Which methods are suitable for preparing both zinc sulfate and copper(II) sulfate?(1)Reacting the metal oxide with warm dilute aqueous sulfuric acid(2) Reacting the metal with dilute aqueous sulfuric acid(3)Reacting the metal
![Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/05/mq2-3.jpg?w=640)
Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition