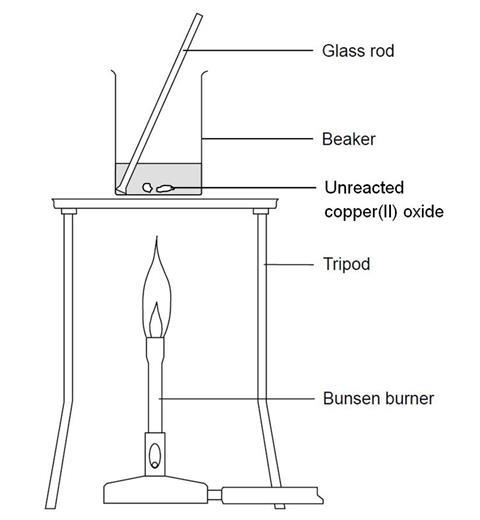
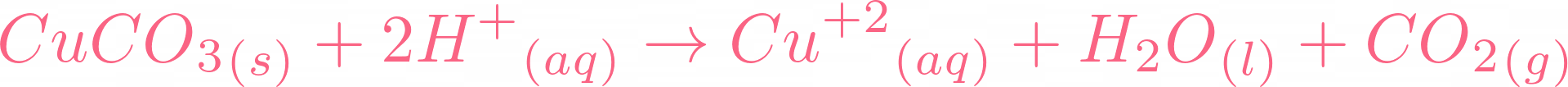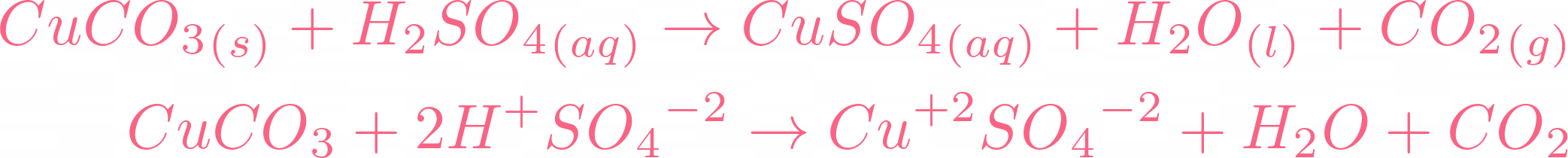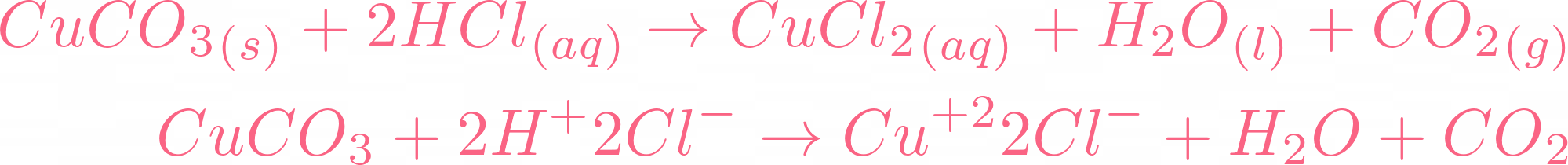copper oxide and sulphuric acid form copper sulphate solution which is filtered to remove unreacted copper oxide see ANA141 Stock Photo - Alamy
1. Last week's homework - mark your work. 2. This week's note – copy or print and glue into notes jotter. 3. This week's

Copper Carbonate,Senior Chemistry - Extended Experimental In-Industry News-Nickel Acetate,Cobalt Sulfate-Fairsky Industrial Co., Limited

Q3. A student wanted to make blue copper sulfate crystals from green copper carbonate powder and sulfuric - Brainly.com

Choosing only substances from the list given in the box below, write equations for the reactions which you would use in the laboratory to obtain: Copper sulphate Dilute sulphuric acid Copper Copper carbonate

Choosing the substances from the list given below, write balanced chemical equations for the reaction which would be used in the laboratory to obtain the following salts. [Dilute sulphuric acid, Copper, Copper(II)
i) Lead sulphate from lead carbonate (ii) Sodium sulphate using dilute sulphuric acid. (iii) Copper chloride using copper carbonate. - Sarthaks eConnect | Largest Online Education Community

Practical: Prepare Copper(II)Sulfate (2.7.8) | Edexcel IGCSE Chemistry Revision Notes 2019 | Save My Exams