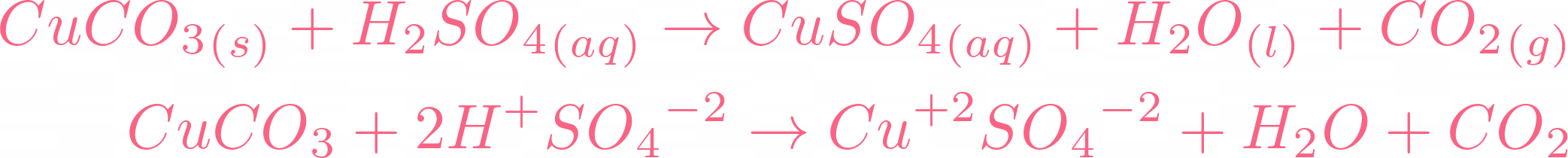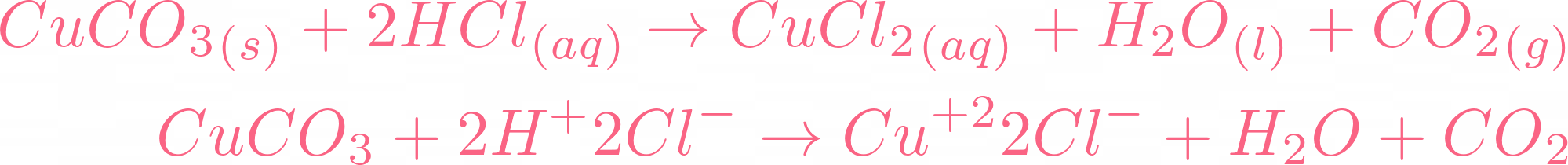i) Lead sulphate from lead carbonate (ii) Sodium sulphate using dilute sulphuric acid. (iii) Copper chloride using copper carbonate. - Sarthaks eConnect | Largest Online Education Community

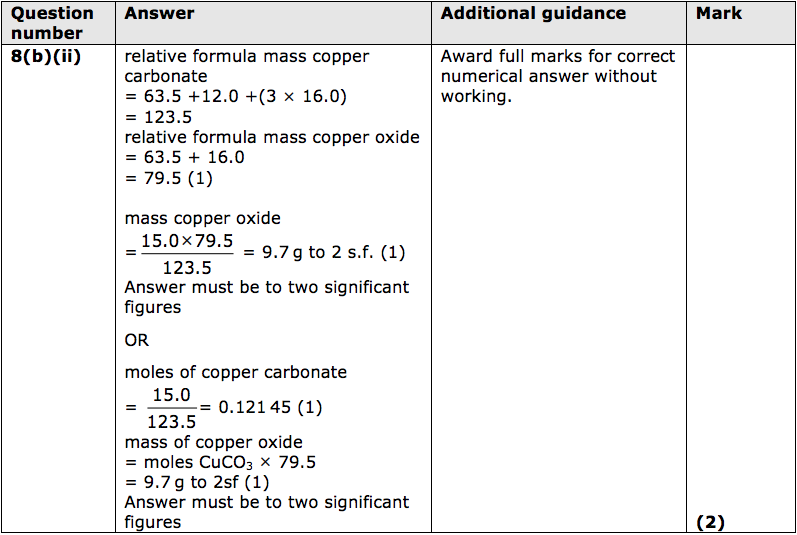
SOLVED: 9.Copper carbonate decomposes to give copper oxide and carbon dioxide a Write a balanced equation for the reaction bWhat mass of copper carbonate is needed to produce 8g of copper oxide?