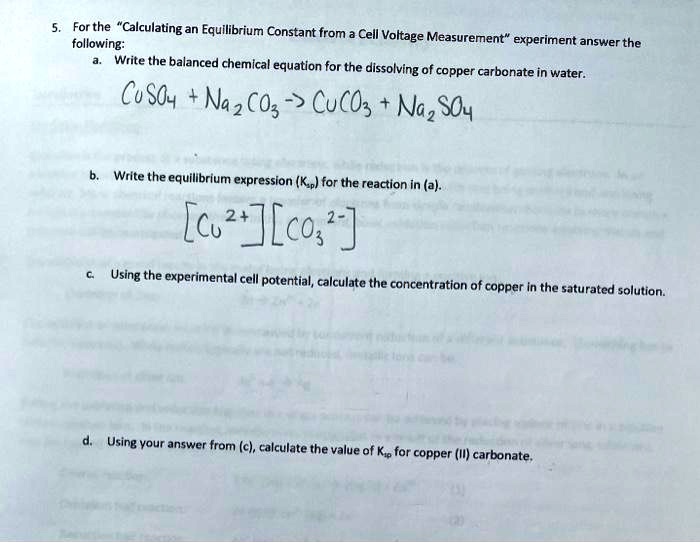Q3 Give reasons why the following are considered as chemical changes 1 Copper Carbonate on heat give...
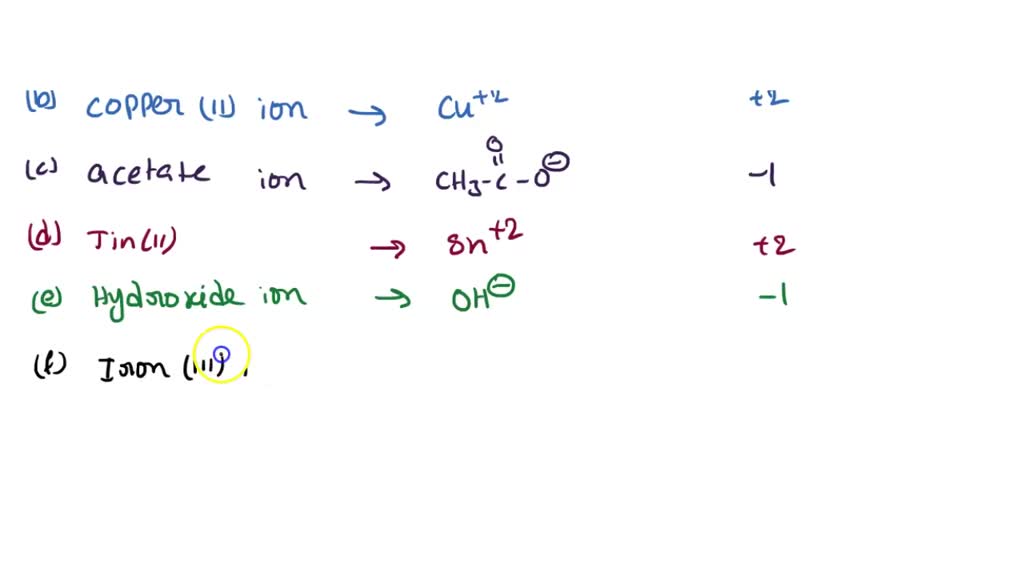
SOLVED: Write the formula and charge for each of the following ions: a. ammonium ion g. copper(II) ion b. acetate ion h. tin(II) ion c. hydroxide ion i. iron(III) ion d. carbonate