Carbon Dioxide from Thermal Decomposition (2.3.4) | Edexcel IGCSE Chemistry Revision Notes 2019 | Save My Exams
![SOLVED: Calculate the mass of carbon dioxide released when 113.100 g of copper carbonate [molar mass = 123.5 g/mol] is heated (assuming that the reaction goes to completion). The molar mass of SOLVED: Calculate the mass of carbon dioxide released when 113.100 g of copper carbonate [molar mass = 123.5 g/mol] is heated (assuming that the reaction goes to completion). The molar mass of](https://cdn.numerade.com/ask_previews/79713408-f4bd-4c6e-a547-0fa321f5c844_large.jpg)
SOLVED: Calculate the mass of carbon dioxide released when 113.100 g of copper carbonate [molar mass = 123.5 g/mol] is heated (assuming that the reaction goes to completion). The molar mass of

Pls solve (ii) Copper (II) carbonate reacts with FISO, to give copper sulphate, carbondioxide and water (i) Write - Chemistry - Some Basic Concepts of Chemistry - 13427939 | Meritnation.com
![Calculate the percentage loss of mass of hydrated copper [II] sulphate [ CuSO4] when it is completely dehydrated. [ CuSO4· 5H2O→ CuSO4 + 5H2O ][At. wts. are Cu = 64, S = 32, O = 16, H = 1]. Calculate the percentage loss of mass of hydrated copper [II] sulphate [ CuSO4] when it is completely dehydrated. [ CuSO4· 5H2O→ CuSO4 + 5H2O ][At. wts. are Cu = 64, S = 32, O = 16, H = 1].](https://dwes9vv9u0550.cloudfront.net/images/10327976/38baafa1-9248-4411-bb94-21fa0ee08540.jpg)
Calculate the percentage loss of mass of hydrated copper [II] sulphate [ CuSO4] when it is completely dehydrated. [ CuSO4· 5H2O→ CuSO4 + 5H2O ][At. wts. are Cu = 64, S = 32, O = 16, H = 1].

Particle size distribution of basic copper carbonate with Na 2 CO 3... | Download Scientific Diagram

HOW MUCH COPPER IS OBTAINED FROM 100gOF COPPER CARBONATE CUCO3, HOW MANY MOLES OF C02 GET BY THERMAL DECOMPOSITION OF - Chemistry - Basic Concepts in Chemistry - 12875641 | Meritnation.com
![SOLVED: Calculate the mass of carbon dioxide released when 201.600 g of copper carbonate [molar mass of 123.5 g/mol] is heated (assuming that the reaction goes to completeion) the molar mass of SOLVED: Calculate the mass of carbon dioxide released when 201.600 g of copper carbonate [molar mass of 123.5 g/mol] is heated (assuming that the reaction goes to completeion) the molar mass of](https://cdn.numerade.com/ask_previews/f32d746-c4b-a5ae-1526-c0d60e835da0_large.jpg)
SOLVED: Calculate the mass of carbon dioxide released when 201.600 g of copper carbonate [molar mass of 123.5 g/mol] is heated (assuming that the reaction goes to completeion) the molar mass of







![Calculate the mass of 100 molecules of copper carbonate. [Atomic mass of Cu = 63.5 u] - Brainly.in Calculate the mass of 100 molecules of copper carbonate. [Atomic mass of Cu = 63.5 u] - Brainly.in](https://hi-static.z-dn.net/files/d0c/83e0ec0d0f383ca7189ba30bed59455d.jpg)





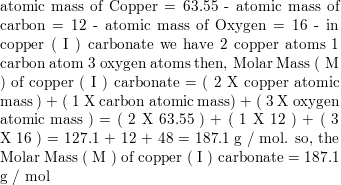
![Calculate the mass of 100 molecules of copper carbonate. [Atomic mass of Cu = 63.5 u] - Brainly.in Calculate the mass of 100 molecules of copper carbonate. [Atomic mass of Cu = 63.5 u] - Brainly.in](https://hi-static.z-dn.net/files/d43/05749e7c0eb98396e5cf28d68efd6f51.jpg)