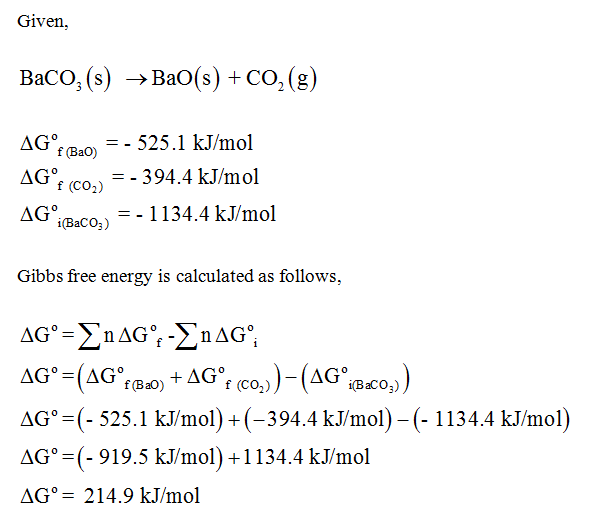Chemical Reactions: Predicting Products and Balancing This PowerPoint presentation will be FAR more effective if you view it as a slideshow This PowerPoint. - ppt download

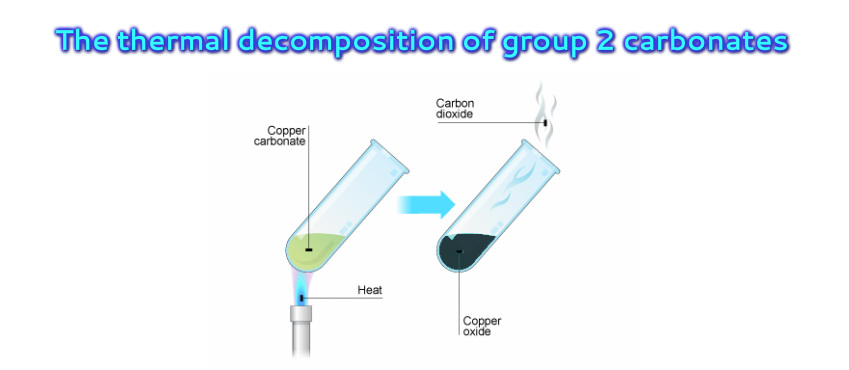
Barium carbonate decomposes when heated. BaCO3(s) → BaO(s) + CO2(g) (a) A student heated a 10.0 g sample of barium carbonate until it was fully decomposed.Calculate the volume of carbon dioxide gas
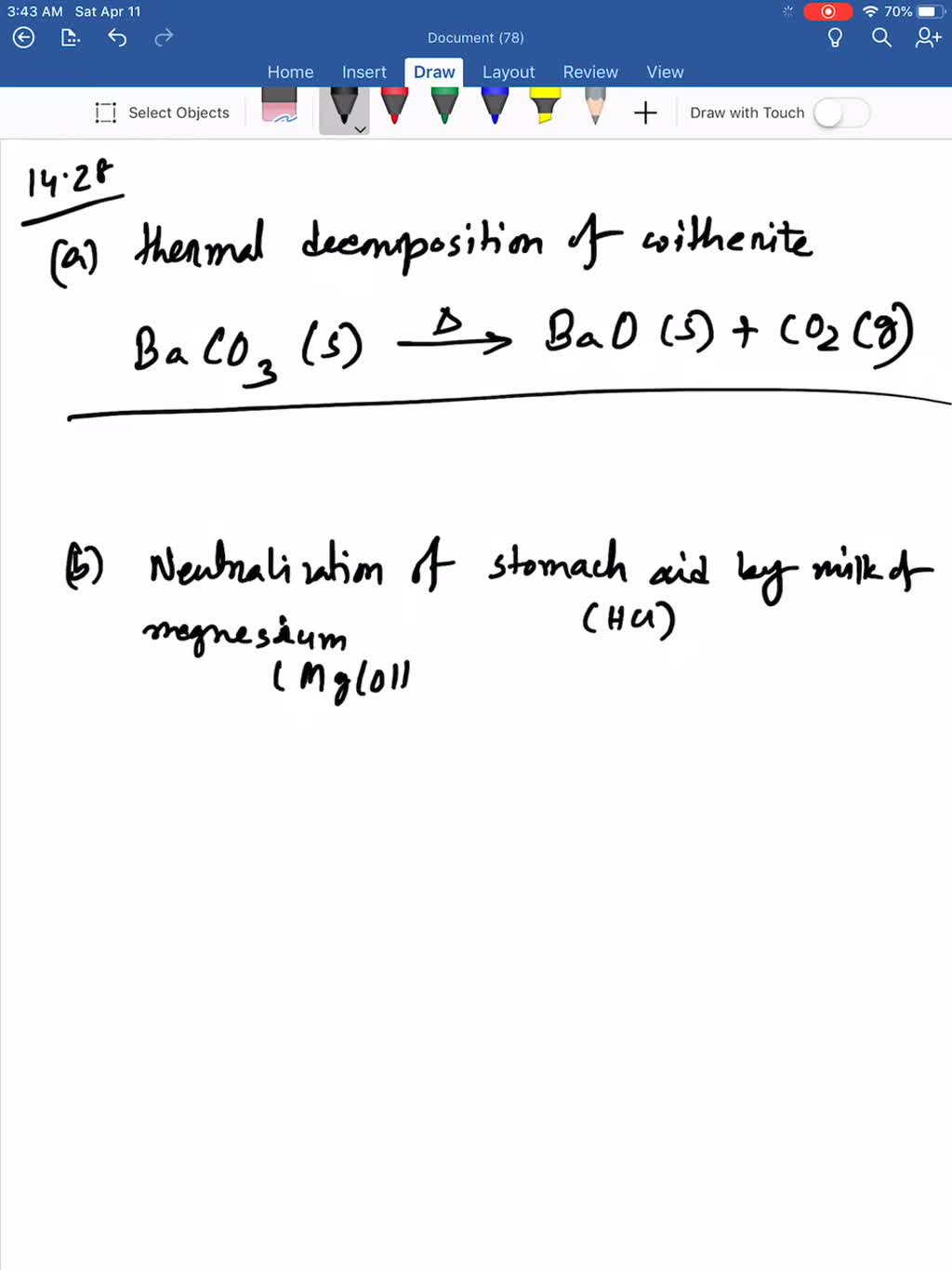
SOLVED:Write a balanced equation for each reaction: (a) Thermal decomposition of witherite (barium carbonate) (b) Neutralization of stomach acid (HCl) by milk of magnesia (magnesium hydroxide)

Formation of Metastable Calcite-type Barium Carbonate During Low-temperature Decomposition of (Ba,Ti)- Precursor Complexes | Afribary
Elaboration and characterization of barium titanate powders obtained by the mechanical activation of barium nitrate and titanate

The formation of oriented barium carbonate from the decomposition of yttria-doped barium zirconate films - ScienceDirect

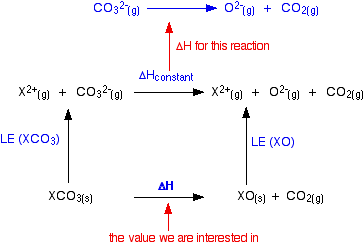
SOLVED: Question #Z (LOmarkshi Consider the decomposition of barium carbonate at standard conditions (below). BaCO3 (s) = BaOc) + COz (g) In what direction will the equilibrium shift ifbarium oxide is added

Barium carbonate decomposes when heated. BaCO3(s) → BaO(s) + CO2(g) (a) A student heated a 10.0 g sample of barium carbonate until it was fully decomposed.Calculate the number of moles of barium

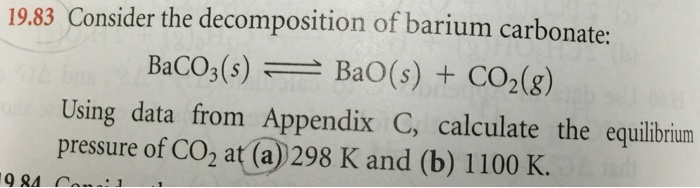





![PDF] Melting curve minimum of barium carbonate BaCO3 near 5 GPa | Semantic Scholar PDF] Melting curve minimum of barium carbonate BaCO3 near 5 GPa | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3de18daeec30ff44d49918f1472776c04f98f970/3-Table1-1.png)