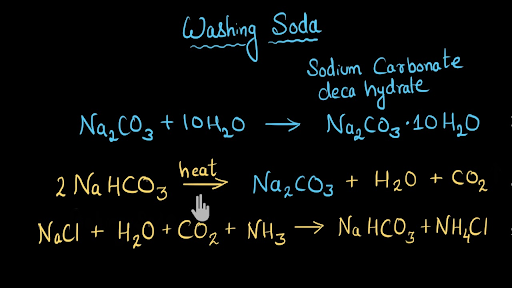
Sodium Carbonate Anhydrous - Sodium Carbonate Anhydrous buyers, suppliers, importers, exporters and manufacturers - Latest price and trends
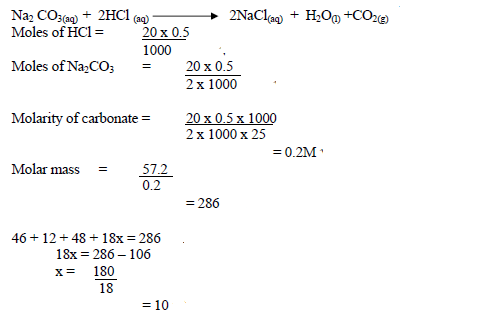
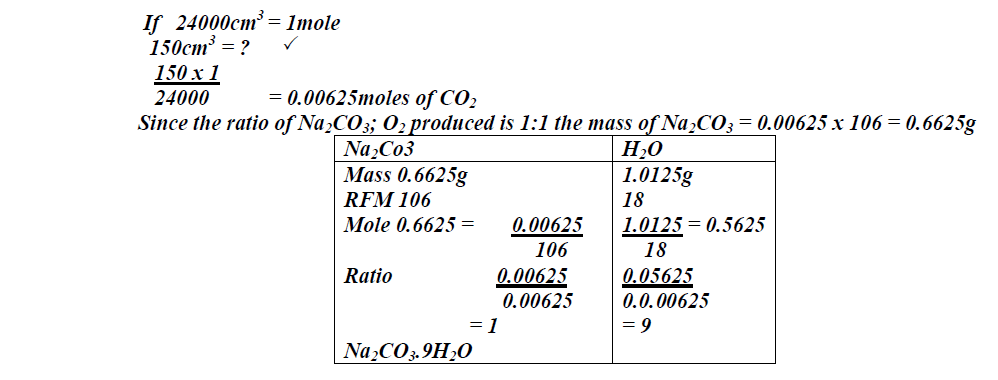
57.2g of hydrated sodium carbonate (Na<sub>2</sub>CO<sub>3</sub>.XH<sub>2</sub>O) were dissolved in water and the solution made to one litre. 20cm<sub>3</sub> of 0.5M HCl recated with 25cm<sub>3</sub> of...
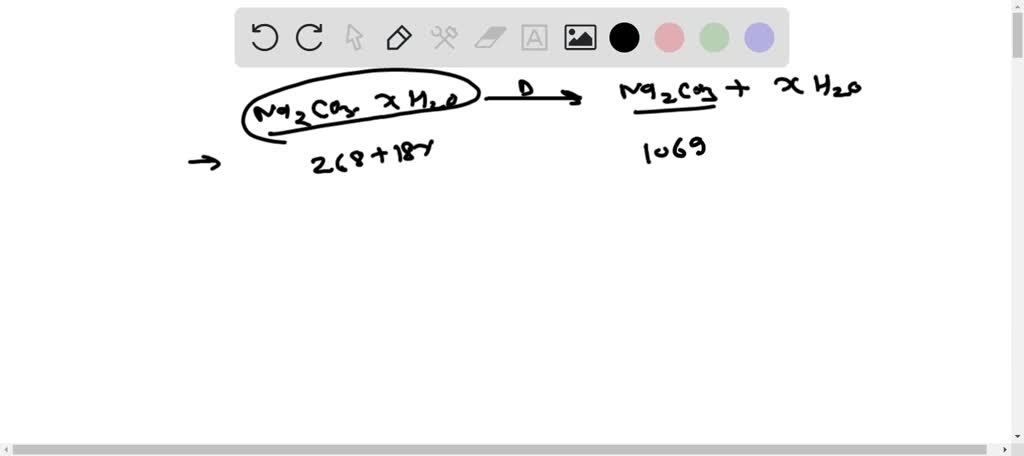
SOLVED: The molar mass of hydrated sodium carbonate is 268g.mol Calculate the number of moles of water of crystalisation (x)If the formula hydrated sodium carbonate is Na2Co3 .xH2O

DOC) To determine the mass of water in hydrated sodium carbonate salt and the value of x in Na2CO3•xH2O | Josephine Yeh - Academia.edu

Sodium Carbonate Stock Illustrations – 106 Sodium Carbonate Stock Illustrations, Vectors & Clipart - Dreamstime
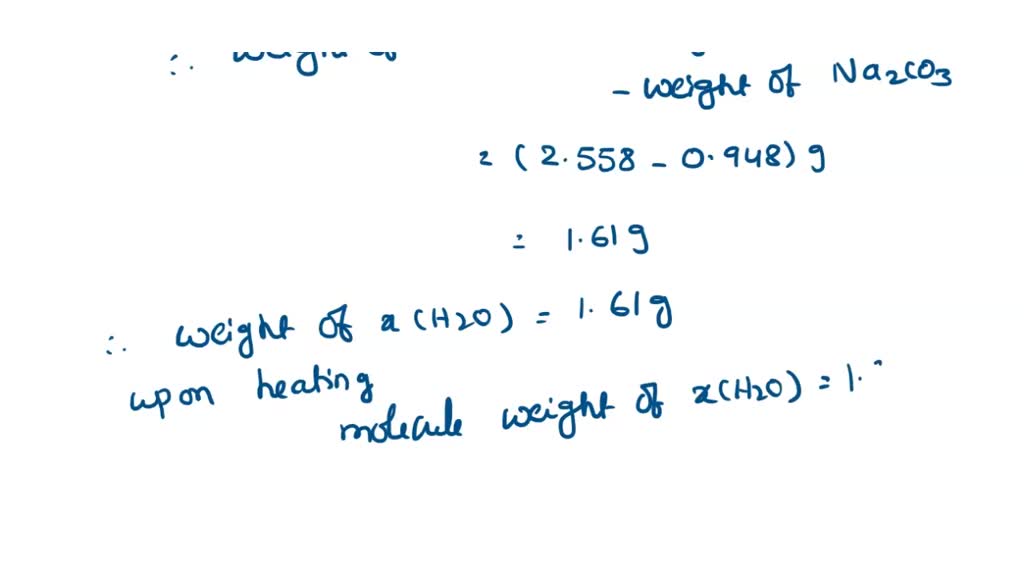
SOLVED: a 2.558 g sample of sodium carbonate hydrate was heated to drive off the water resulting in 0.948 g of anhydrous sodium carbonate. Calculate the formula of the hydrate.